Advertisements
Advertisements
Question
Use the molecular orbital energy level diagram to show that \[\ce{N2}\] would be expected to have a triple bond, \[\ce{F2}\], a single bond and \[\ce{Ne2}\], no bond.
Solution
Formation of N2 molecules.
Electrons configuration of N-tom .7 N = `1s^2, 2s^2, 2p_x^1, 2p_y^1, 2p_z^1`,N2 molecules = `sigma1s^2, sigma^∗1s^2, sigma2s^2, pi2p_x^2 = pi2p_y^2, pi2p_z^2`
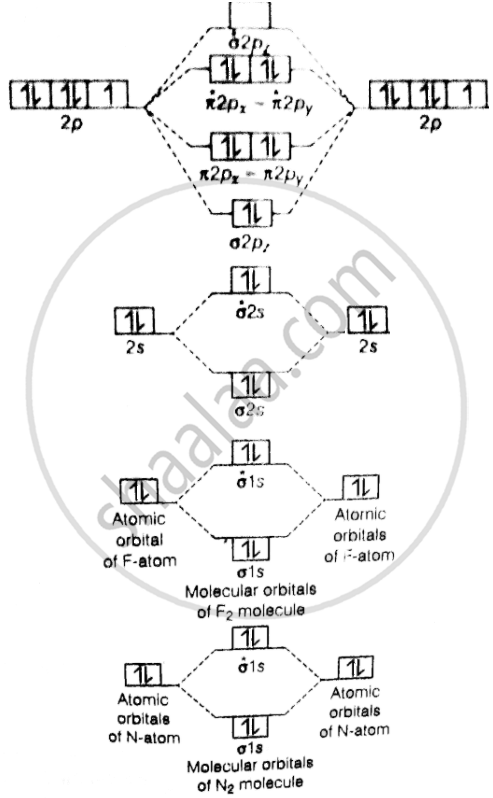
Bond order = `1/2[N_b - N_a] = 1/2(10 - 4)` = 3.
Bond order value of 3 means that N2 contains a triple bond.
Formation of F2 molecule, .9 F = `1s^2, 2s^2, 2p_x^2, sigma^∗2s^2, sigma2p_z^2, pi2p_x^2, pi2p_x^2 = pi2p_y^2, pi^∗2p_z^2 = pi^∗2p_y^2`

Bond order = `1/2[N_b - N_a] = 1/2(10 - 8)` = 1
Bond order value 1 means that \[\ce{F2}\] contains single bond.
Formation of Ne2 molecule 10 Ne = `1s^2, 2p_x^2, 2p_y^2, 2p_z^2`
Ne molecule = `sigma1s^2, sigma^∗1s^2, sigma2s^2, sigma^∗2s^2, sigma2p_z^2, pi2p_z^2, = pi2p_y^2, pi^∗2p_x^2 = pi^∗2p_y^2, sigma^∗2p_z^2`
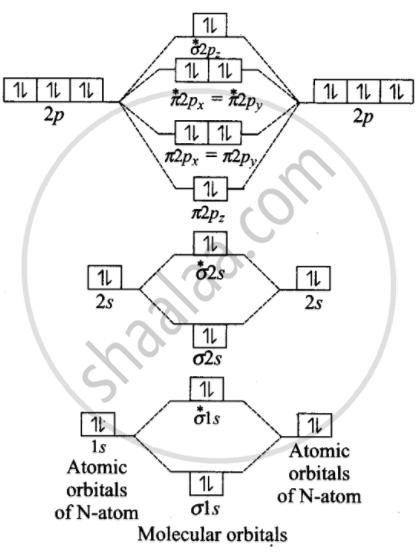
Bond order = `1/2[N_b - N_a] = 1/2(10 - 10)` = 0
Bond order value zero means that there is no formation of bond between two Ne-atoms. Hence, Ne2 molecole does not exist.
