Advertisements
Advertisements
Question
Briefly describe the valence bond theory of covalent bond formation by taking an example of hydrogen. How can you interpret energy changes taking place in the formation of dihydrogen?
Solution
The valence bond theory was put forward by Heitler and London in 1927. It was later improved and developed by L. Pauling and J.C. Slater in 1931. The valence bond theory is based on the knowledge of atomic orbitals and electronic configurations of elements, overlap criteria of atomic orbitals and stability of molecule.
The main points of valence bond theory are
(i) Atoms do not lose their identity even after the formation of the molecule.
(ii) The bond is formed due to the interaction of only the valence electrons as the two atoms come close to each other. The inner electrons do not participate in the bond formation.
(iii) During the formation of bond, only the valence electrons from each bonded atom lose their identity. The other electrons remain unaffected.
(iv) The stability of bond is accounted by the fact that the formation of bond is accompanied by release of energy. The molecule has minimum energy at a certain distance between the atoms known as intemuclear distance. Larger the decrease in energy, stronger will be the bond formed.
Valence bond Treatment of Hydrogen Molecule: Consider two hydrogen atoms A and B approaching each other having nuclei Ha and HB and the corresponding electrons eA and eB respectively.
When atoms come closer to form molecules new forces begin to operate.
(a) The force of attraction between nucleus of atom and electron of another atom.
(b) The force of repulsion between two nuclei of the atom and electron of two atoms.

Fig. (a) Two hydrogen atoms at a large distance and hence, no interaction, (b) Two hydrogen atom closer to each other atomic orbitals begin to interact, (c) Attractive and repulsive forces in hydrogen atoms when interaction begins. In case of hydrogen: Figure ‘a’ shows that two hydrogen atoms are at farthest distances and their electron distribution is absolutely symmetrical.
(a) When two hydrogen atom start coming closer to each other, the electron cloud becomes distorted and new attractive and repulsive forces begin to operate as shown in figure ‘c’
(b) In figure ‘c’ dotted lines show attractive forces present in atom already and bold lines show the new attractive and repulsive forces.
(c) It has been found experimentally that the magnitude of net attractive forces is more than net repulsive forces. Thus stable hydrogen molecule is formed.
Potential energy diagram for formation of hydrogen molecules: When two hydrogen atoms are at farther distance, there is no force operating between them, when they start coming closer to each other, force of attraction comes into play and their potential energy starts decreasing. As they come closer to each other potential goes on decreasing, but a point is reached, when potential energy acquires minimum value. Note:
(a) This distance corresponding to this minimum energy value is called the distance of maximum possible approach, i.e. the point which corresponds to minimum energy and maximum stability.
(b) If atoms come further closer than this distance of maximum possible approach, then potential energy starts increasing and force of repulsion comes into play and molecules starts becoming unstable.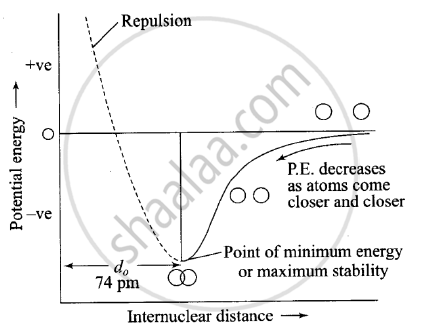
Fig. Variation of potential energy of interaction between two hydrogen atoms.
APPEARS IN
RELATED QUESTIONS
Draw an orbital diagram of Fluorine molecule
Explain geometry of methane molecule on the basis of Hybridization.
Give a reason for the sigma (σ) bond is stronger than the Pi (π) bond.
Give the type of overlap by which the pi (π) bond is formed.
When ones and three p orbitals hybridise,
Ethene molecule has ____________ sp2 -s σ bond(s), ____________ sp2 -sp2 σ bond(s) and ____________ p-p π bond(s).
The number of sigma bonds in vanillin is ____________.
If the electronic configuration of an element is 1s2 2s2 2p6 3s2 3p6 3d2 4s2, the four electrons involved in chemical bond formation will be ______.
Which of the following statements is INCORRECT according to the valence bond theory?
The \[\ce{H - N - H}\] bond angle in ammonia molecule is ______.
