Advertisements
Advertisements
Question
If the electronic configuration of an element is 1s2 2s2 2p6 3s2 3p6 3d2 4s2, the four electrons involved in chemical bond formation will be ______.
Options
3p6
3p6, 4s2
3p6, 3d2
3d2, 4s2
Solution
If the electronic configuration of an element is 1s2 2s2 2p6 3s2 3p6 3d2 4s2, the four electrons involved in chemical bond formation will be 3d2, 4s2.
Explanation:
The electrons involved in the formation of any chemical bond are the valence shell electrons. In the given electronic configuration 1s2 2s2 2p6 3s2 3p6 3d2 4s2 , the valence shell electrons are in d-orbital and s-orbital.
APPEARS IN
RELATED QUESTIONS
Explain the formation of H2 molecule on the basis of valence bond theory.
Distinguish between sigma and pi bond.
Give a reason for carbon is tetravalent in nature.
Mention the steps involved in Hybridization.
Which one of the following is the likely bond angles of sulphur tetrafluoride molecule?
In ClF3, NF3 and BF3 molecules the chlorine, nitrogen and boron atoms are ______.
The correct order of O – O bond length in hydrogen peroxide, ozone and oxygen is
Among the following, the compound that contains, ionic, covalent and Coordinate linkage is ______.
What is a pi - bond?
Why does type of overlap given in the following figure not result in bond formation?
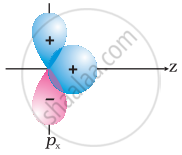 |
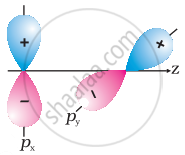 |
