Advertisements
Advertisements
Question
Give the type of overlap by which the pi (π) bond is formed.
Solution
The type of overlap by which the pi (π) bond is formed is p–p lateral overlap.
APPEARS IN
RELATED QUESTIONS
Explain the formation of H2 molecule on the basis of valence bond theory.
Give reasons for need of Hybridization
Explain geometry of methane molecule on the basis of Hybridization.
Give a reason for carbon is tetravalent in nature.
Identify the type of overlap present in H2. Explain diagrammatically.
Complete the following Table.
| Molecule | Type of Hybridization | Type of bonds | Geometry | Bond angle |
| CH4 | - | 4C-H 4σ bonds |
Tetrahedral | - |
| NH3 | sp3 | 3N-H 3σ bonds 1 lone pair |
- | - |
| H2O | - | - | angular | 104.5° |
| BF3 | sp2 | - | - | 120° |
| C2H4 | - | - | - | 120° |
| BeF2 | - | 2 Be-F | Linear | - |
| C2H2 | sp | (3σ+2π) 1C-C σ 2C-H σ 2C-C π |
- | - |
According to Valence bond theory, a bond between two atoms is formed when ______.
In ClF3, NF3 and BF3 molecules the chlorine, nitrogen and boron atoms are ______.
Which of these represents the correct order of their increasing bond order.
The correct order of O – O bond length in hydrogen peroxide, ozone and oxygen is
XeF2 is isostructural with ______.
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
1s and 2py
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2py
Considering x-axis as the molecular axis which out of the following will form a sigma bond.
2px and 2pz
Ethene molecule has ____________ sp2 -s σ bond(s), ____________ sp2 -sp2 σ bond(s) and ____________ p-p π bond(s).
Which of the following is correct decreasing order of the repulsive Interaction of electron pairs in a molecule?
The overlap of orbitals involved in the formation of C - Br bond in vinyl bromide is ______.
Why does type of overlap given in the following figure not result in bond formation?
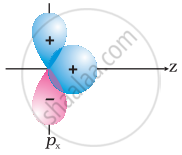 |
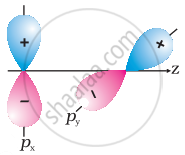 |
Briefly describe the valence bond theory of covalent bond formation by taking an example of hydrogen. How can you interpret energy changes taking place in the formation of dihydrogen?
Match List - I with List - II.
| List - I | List - II | ||
| (a) | \[\ce{PCl5}\] | (i) | Square pyramidal |
| (b) | \[\ce{SF6}\] | (ii) | Trigonal planar |
| (c) | \[\ce{BrF5}\] | (iii) | Octahedral |
| (d) | \[\ce{BF3}\] | (iv) | Trigonal bipyramidal |
Choose the correct answer from the options given below.
