Advertisements
Advertisements
Question
Describe hybridisation in the case of \[\ce{PCl5}\] and \[\ce{SF6}\]. The axial bonds are longer as compared to equatorial bonds in \[\ce{PCl5}\] whereas in \[\ce{SF6}\] both axial bonds and equatorial bonds have the same bond length. Explain.
Solution
Hybridisation in the case of \[\ce{PCl5}\]: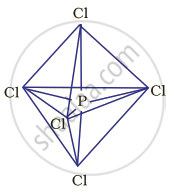
The hybridisation of phosphorus \[\ce{PCl5}\] is sp3d and it has a trigonal bipyramidal geometry. The axial bonds in \[\ce{PCl5}\] are slightly longer as compared to the equatorial bonds because axial bonds experience greater repulsive forces from other bonds as compared to the equatorial bonds.
Hybridisation in the case of \[\ce{SF6}\]: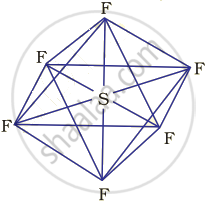
The hybridisation of sulphur in \[\ce{SF6}\] is sp3d2 and the molecule has an octahedral geometry. The bond length of axial as well as the equatorial bonds are similar because all the bonds in octahedral geometry experience similar repulsive forces from other bonds.
APPEARS IN
RELATED QUESTIONS
Define the bond length.
In which of the following molecule/ion all the bonds are not equal?
Which of the following statements are correct about \[\ce{CO^{2-}3}\]?
(i) The hybridisation of central atom is sp3.
(ii) Its resonance structure has one \[\ce{C - O}\] single bond and two \[\ce{C = O}\] double bonds.
(iii) The average formal charge on each oxygen atom is 0.67 units.
(iv) All \[\ce{C - O}\] bond lengths are equal.
All the \[\ce{C - O}\] bonds in carbonate ion \[\ce{(CO^{2-}3)}\] are equal in length. Explain.
In a covalent bond, ______ the bond length ______ is the energy required to break the bond.
