Advertisements
Advertisements
Question
What does the diffusion of gases tell us about their particles?
Solution
The diffusion of gases tells us that:
- Matter consists of tiny particles, that have space between them.
- The particles of matter are in constant motion.
APPEARS IN
RELATED QUESTIONS
Give reasons:
A gas exerts pressure on the walls of the container.
Give a reason for the following observation.
We can get the smell of perfume sitting several metres away.
Name two gases which are supplied in compressed form in homes and hospitals.
Name the three states of matter. Give one example of each.
Explain how, the smell of food being cooked in the kitchen reaches us even from a considerable distance.
Explain why, diffusion occurs more quickly in a gas than in a liquid.
Name two gases of air which dissolve in water by diffusion. What is the importance of this process in nature?
A student placed a gas jar containing air in the upside down position over a gas jar full of red-brown bromine vapours. He observed that the red-brown colour spread upwards into the jar containing air. Based on this observation, the student concluded that it is only the bromine vapour which moves up and diffuses into air in the upper jar, the air from the upper jar does not move down by diffusion into the lower jar containing bromine vapours. Do you agree with this conclusion of the student? Give reason for your answer.
Explain why?
- A gas fills a vessel completely?
- Camphor disappears without leaving any residue.
 |
|
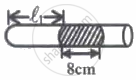
| Fig. I | Fig. II |
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)