Advertisements
Advertisements
Question
What happens when (NH4)2Cr2O7 is heated? Write the equations.
Solution
When (NH4)2Cr2O7 is heated its decompose to produces Cr2O3, H2O and N2
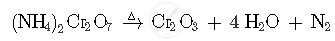
APPEARS IN
RELATED QUESTIONS
What is the action of Excess of air on ammonia ?
What is the action of the following reagents on ammonia :
Nessler's reagent
Mention the conditions required to maximise the yield of ammonia.
How does ammonia react with a solution of Cu2+?
What is the action of Excess of chlorine on ammonia?
In laboratory ammonia is prepared by heating:
Liquid ammonia bottles are opened after cooling them in ice for some time. It is because liquid NH3 ____________.
Ammonia is generally manufactured for fertilizers by the reaction:
Which one of the following is not a use of ammonia?
In the preparation of HNO3, we get NO gas by catalytic oxidation of ammonia. The moles of NO produced by the oxidation of two moles of NH3 will be ______.
