Advertisements
Advertisements
Question
What is the principle of the method of mixtures?
Solution
The principle of the method of mixtures says that the heat lost by a hot body is equal to the heat gained by a cold body.
RELATED QUESTIONS
Why is the base of a cooking pan generally made thick?
Convection is the process by which the thermal energy flows in solids.
Define thermal capacity.
Heat capacity C’= ______.
Water is used as a coolant because it ______.
Compare heat capacity and specific heat capacity.
Thermal capacities of substances A and B are same. If mass of A is more than mass of B then:
Which substance will show greater rise in temperature if the same amount of heat is supplied to both?
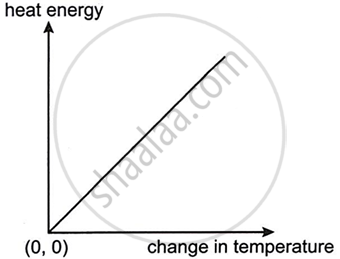
The graph given below shows heat energy supplied against change in temperature when no energy is lost to the surroundings. The slope of this graph will give:
A solid of mass 60 g at 100°C is placed in 150 g of water at 20°C. The final steady temperature is 25°C. Calculate the heat capacity of solid.
[sp. heat capacity of water = 4.2 J g-1 K-1]
- Calculate the heat capacity of a copper vessel of mass 200 g if the specific heat capacity of copper is 410 J kg-1 K-1.
- How much heat energy will be required to increase the temperature of the vessel in part (a) from 25°C to 35°C?
