Advertisements
Advertisements
Question
What is the significance of leaching in the extraction of aluminium?
Solution
In the extraction of aluminium, the significance of leaching is to concentrate pure alumina (Al2O3) from bauxite ore.
Bauxite usually contains silica, iron oxide, and titanium oxide as impurities. In the process of leaching, alumina is concentrated by digesting the powdered ore with a concentrated solution of NaOH at 473-523 K and 35-36 bar. Under these conditions, alumina (Al2O3) dissolves as sodium meta-aluminate and silica (SiO2) dissolves as sodium silicate leaving the impurities behind.
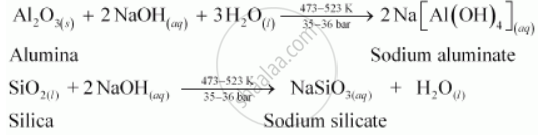
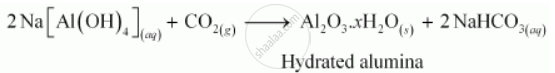
The impurities are then filtered and the solution is neutralized by passing CO2 gas. In this process, hydrated Al2O3 gets precipitated and sodium silicate remains in the solution. Precipitation is induced by seeding the solution with freshly prepared samples of hydrated Al2O3.
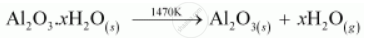
Hydrated alumina thus obtained is filtered, dried, and heated to give back pure alumina (Al2O3).
APPEARS IN
RELATED QUESTIONS
How can you separate alumina from silica in bauxite ore associated with silica? Give equations, if any
How is leaching carried out in case of low grade copper ores?
Write the chemical reactions involved in the process of extraction of Gold. Explain the role of dilute NaCN and Zn in this process.
What is the role of dilute NaCN in the extraction of gold ?
Which reducing agent is employed to get copper from the leached low-grade copper ore?
The common impurity present in most of the ores is ____________.
Common impurities present in bauxite are:
(i) \[\ce{CuO}\]
(ii) \[\ce{ZnO}\]
(iii) \[\ce{Fe2O3}\]
(iv) \[\ce{SiO2}\]
The process of converting hydrated alumina into anhydrous alumina is called
In Baeyer's process, the leaching of alumina is done by using ______
Gold is extracted by making soluble cyanide complex. The cyanide complex is ______.
Which of the reaction is suitable for concentrating ore by leaching process?
