Advertisements
Advertisements
Question
When kept in water, raisin swells in size. Name and explain the phenomenon involved with the help of a diagram. Give three applications of the phenomenon.
Solution
When kept in water, raisen swells in size. It is because of a phenomenon known as 'Osmosis'. THis process is diagrametically represented in figure below.. In this process a solution is separated from solvent by a semi permeable membrane which allows the passage of solvent molecules but does not permit solute particles to pass through..
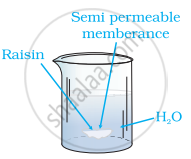
Osmosis is the process of flow of solvent molecules from pure solvent to the solution through semi permeable membrance.
Three applications of osmosis area:
(i) Movement of water from soil into plant roots and subsequently iinto upper portion of the plant is partly due to osmosis.
(ii) Preservation of meat against bacterial action by adding salt.
(iii) Preservation of fruits against bacterial action by adding sugar. Bacterium in canned fruit loses water through the process of osmosis, shrivels and dies.
APPEARS IN
RELATED QUESTIONS
Which of the two components of starch is water soluble?
An unripe mango placed in a concentrated salt solution to prepare pickle, shrivels because ______.
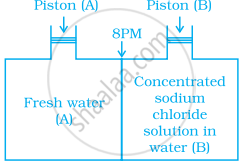
Consider the figure and mark the correct option.
Water retention or puffiness due to high salt intake occurs due to ______.
Why are aquatic species more comfortable in cold water in comparison to warm water?
Evaporation of water is?
Molal enthalpy of fusion of water at 273 k is 6.0246 kg mol–1 calculated molal depression constant.
