Advertisements
Advertisements
Question
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Options
Reaction of nitrite with \[\ce{LiAlH4}\].
Reaction of amide with \[\ce{LiAlH4}\] followed by treatment with water
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis.
Treatment of amide with bromine in aqueous solution of sodium hydroxide.
Solution
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis.
Explanation:
Aliphatic and arylalkyl primary amines can be prepared by the reduction of the corresponding nitriles with \[\ce{LiAlH4}\].
\[\ce{\underset{Alkynitrite}{R - C ≡ N} \underset{Arynitrite}{Ar - C ≡ N} -> LiAlH4 RCH2\underset{1° amine}{NH2} or ArCh2NH2}\]
Heating alkyl halide with primary, secondary and tertiary amine can be prepared by reduction of \[\ce{LiAlH4}\] followed by treatment with water.
\[\ce{\underset{1° amide}{R - CONH2} ->[(i) LiAlH4][(ii) H2O] R - CH2 - NH2}\]
Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis produces primary amine. This process is known as Gabriel phthalimide reaction. The number of carbon atoms in the chain of amines of product is same as reactant.
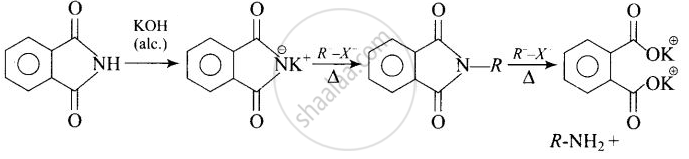
APPEARS IN
RELATED QUESTIONS
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH + Br2] C}\]
Give the structures of A, B and C in the following reactions :
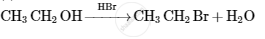
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
Which of the following does NOT give carbylamine test?
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
How will you carry out the following conversions?
Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Write a short note on the following:
Ammonolysis.
