Advertisements
Advertisements
Question
Why is water molecule polar?
Short Note
Solution
Water molecules are polar because in the gas phase water is a bent molecule with a bond angle of 104.5°, \[\ce{O - H}\] bond length of 95.7 pm.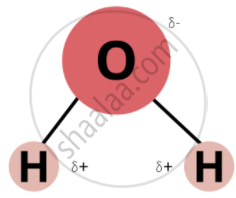
shaalaa.com
Water - Structure of Water
Is there an error in this question or solution?
