Advertisements
Advertisements
Question
Why does water show high boiling point as compared to hydrogen sulphide? Give reasons for your answer.
Solution
Because of hydrogen bonding water has a higher boiling point than \[\ce{H2S}\] as in \[\ce{H2S}\] hydrogen bonding is absent.
APPEARS IN
RELATED QUESTIONS
Arrange the following:
CaH2, BeH2 and TiH2 in order of increasing electrical conductance.
Describe the usefulness of water in biosphere and biological systems.
What properties of water make it useful as a solvent? What types of compound can it (i) dissolve, and (ii) hydrolyse?
Why is it said that-
Water is a universal solvent.
Explain the picture in your own words.
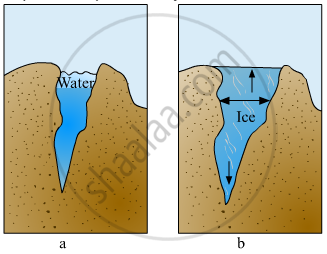
Give reasons: Lakes freeze from top towards bottom.
Explain why \[\ce{HCl}\] is a gas and \[\ce{HF}\] is a liquid.
The freezing point of water ______ with an increase in pressure.
One gram of water requires ______ of heat to raise its temperature by l°C.
Water is circulated around the car engine using the ______ pump and the heat is absorbed.
