Advertisements
Advertisements
Question
Work out the formulae for the following compounds :
Sodium oxide
Solution
Valency of Sodium = 1
Valency of Oxygen = 2
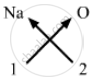
Formula for sodium oxide can be worked out by crossing the valencies of the constituent elements.
Therefore, formula for sodium oxide is: Na2O.
APPEARS IN
RELATED QUESTIONS
Name the scientist who gave law of constant proportions.
Which postulate of Dalton's atomic theory is the result of law of conservation of mass given by Lavoisier ?
The law of constant proportions was given by :
In an experiment, 4.90 g of copper oxide was obtained from 3.92 g of copper. In another experiment, 4.55 g of copper oxide gave, on reduction, 3.64 g of copper. Show with the help of calculations that these figures verify the law of constant proportions.
When 5 g of calcium is burnt in 2 g of oxygen, then 7 g of calcium oxide is produced. What mass of calcium oxide will be produced when 5 g of calcium is burnt in 20 g of oxygen ? Which law of chemical combination will govern your answer ?
What do we call those particles which have more or less electrons than the normal atoms ?
What do we call those particles which have less electrons than the normal atoms ?
The same proportion of carbon and oxygen in the carbon dioxide obtained from different sources proves the law of ______.
State the law of constant proportions.
Ruthresh, Hari, Kanishka, and Thahera collected different samples of water from a well, a pond, a river, and underground water. All these samples were sent to a testing laboratory. The test result showed the ratio of hydrogen to oxygen as 1:8.
- What conclusion would you draw from the above experiment?
- Which law of chemical combination does it obey?
