Advertisements
Advertisements
Question
Write a short note on coupling reactions.
Solution
The reaction involves the retention of diazo groups.
- Arenediazonium salts when treated with certain reactive aromatic compounds such as phenols or aromatic amines, give azo compounds.
- These have extended conjugated system of double bonds in which two aromatic rings are joined through the azo group \[\ce{- N = N -}\]. This reaction is called azo coupling.
- Azo compounds are brightly coloured and are used as dyes.
- This is an example of an electrophilic aromatic substitution reaction. Here, the electrophiles are positively charged diazonium ions.
- Substitution usually occurs para to the ring activating group.
e.g. Benzenediazonium chloride reacts with phenol in mild alkaline medium to give p-hydroxyazobenzene (orange dye).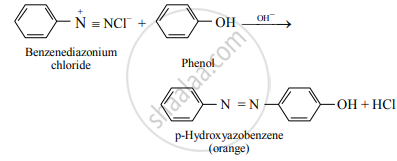
- Azo coupling with β-naphthol in NaOH is used as a confirmatory test for primary aromatic amines.
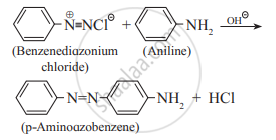
- Benzenediazonium chloride reacts with aniline in mild alkaline medium to give p-aminoazobenzene (yellow dye.)
APPEARS IN
RELATED QUESTIONS
Choose the most correct option.
Which of the following compounds contains azo linkage?
Answer the following
Complete the following reaction :
`"C"_6"H"_5"N"_2^+"Cl"^-` + C2H5OH →
The reaction in which diazonium salt is used
Draw resonance structures of aryl diazonium salts.
Write the use of aryl diazonium salts.
Explain the following reaction with a suitable example.
Sandmeyer reaction
Which of the following reaction of diazonium salt involves retention of diazonium group?
When a quaternary ammonium hydroxide is strongly heated, an alkene and a tertiary amine are formed as the products. This reaction is known as ____________.
The reagent used in Gatterman reaction is ____________.
Benzenediazonium chloride on reaction with 'X' gives benzene. Identify X.
Which of the following reaction does NOT involve replacement of diazonium group?
Replacement of diazo group by Cl using cuprous chloride is known as ____________.
Formation of p-hydroxyazobenzene from benzene diazonium chloride and phenol in mild alkaline medium is a ______.
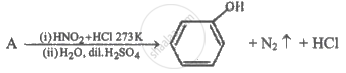
Identify compound 'A' in the following reaction.
What is the product X obtained in the following reaction?
\[\ce{C6H5N2Cl ->[(i) HBF4, (ii) NaNO2/Cu][\Delta] X + N2 + NaBF4}\]
Identify product B in the following reaction.
\[\ce{C6H5N^+_2X^- ->[HBF4] A ->[NaNO2][Cu] B}\]
In the following reaction the reagent A is,
\[\ce{Ar - N^+_2 X^- ->[A] Ar - N^+_2 BF^-_4 ->[\Delta] Ar - F + BF3 + N2 ^}\]
Benzene diazonium chloride on reaction with ethanol forms ____________.
