Advertisements
Advertisements
Question
Write down the formulae of sodium oxide.
Solution
| Ions present | Formula |
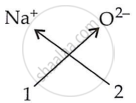 |
Na2O |
APPEARS IN
RELATED QUESTIONS
Write down the formula of aluminium chloride.
The formula of carbonate of a metal M is M2CO3.
What will be the formula of its iodide ?
Complete the following statement by selecting the correct option :
The correct formula of aluminium oxide is
Match the following:
| Column A | Column B |
| (a) Pb(NO3)2 | (i) rotten eggs smell |
| (b) CO2 | (ii) burns with a pop sound |
| (c) (NH4)2Cr2O7 | (iii) the suffocating smell of sulphur |
| (d) HCI | (iv) lime water turns milky |
| (e) NO2 | (v) crackling sound |
| (f) O2 | (vi) residue swells up |
| (g) H2 | (vii) brown gas |
| (i) H2S | (viii) supports combustion |
| (j) SO2 | (ix) fumes with NH3 solution |
Name the element in the compound and give the formula of the following compound:
Ethanol
Write the formula of the following compound:
Potassium bisulphite
Write the formula of the following compound:
Aluminium nitrate
Write the formula of the following compound:
Lead [II] nitrate
Write the formula of the following compound:
Diamine silver chloride
Give the chemical formulae for the following compounds and compute the ratio by mass of the combining elements in each one of them. (You may use appendix-III).
- Ammonia
- Carbon monoxide
- (Hydrogen chloride
- Aluminium fluoride
- Magnesium sulphide
