Advertisements
Advertisements
Question
Write the steps needed for flame test ?
Solution
Flame test :
1. Make a loop at the tip of the platinum wire and dip it in conc. HCl.
2. Put it on the non luminous part of the flame, to see if it gives colour. Repeat the process till it gives no colour to the flame.
3. Prepare a paste of the given salt in a watch glass using conc. HCl.
4. Load the loop of the wire with this prepared paste and introduce it into the non luminous flame of the bunsen burner and then observe the colour of the flame indicating different elements.
APPEARS IN
RELATED QUESTIONS
Name the experiment which demonstrates that hydrogen chloride is soluble in water.
Complete the following reaction and balance them.
Zn+ 2HCl ⟶
Name the gas evolved when dilute hydrochloric acid is added to: Lead (II) sulphide
Give a balanced equation when dilute hydrochloric acid is added to : Calcium carbonate
Give a balanced equation when dilute hydrochloric acid is added to : Potassium bisulphite
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution:
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | _________ | _________ |
| 2. | Magnesium ribbon | _________ | _________ |
| 3. | Manganese (IV) oxide with heating | _________ | _________ |
| 4. | Sodium sulphide | _________ | _________ |
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl ->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
NH4OH + HCl ⟶
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
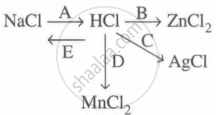
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.