Advertisements
Advertisements
Question
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution:
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | _________ | _________ |
| 2. | Magnesium ribbon | _________ | _________ |
| 3. | Manganese (IV) oxide with heating | _________ | _________ |
| 4. | Sodium sulphide | _________ | _________ |
Solution
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | \[\ce{CO2}\] | Odourless |
| 2. | Magnesium ribbon | \[\ce{H2}\] | Odourless |
| 3. | Manganese (IV) oxide with heating | \[\ce{Cl2}\] | Strong Pungent odour |
| 4. | Sodium sulphide | \[\ce{H2S}\] | Unpleasant like |
RELATED QUESTIONS
Name two gases which chemically combine to form a liquid.
Name the chemical in which gold can be dissolved.
Complete the following reaction and balance them.
Ca(HCO3)2 + 2HCl →
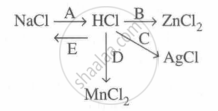
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Give a balanced equation when dilute hydrochloric acid is added to : Zinc Metal
Give a balanced equation when dilute hydrochloric acid is added to : Sodium sulphite
Give a balanced equation when dilute hydrochloric acid is added to : Magnesium bicarbonate
- Give only one suitable chemical test to identify the following gases.
- Ammonia
- Sulphur dioxide
- Hydrogen chloride
- Chlorine
- Carbon dioxide
- Oxygen
- Hydrogen
- Select a basic gas mentioned in Q. (a). How is the basic nature suspected?
- Select acidic gases from the gases mentioned in Q. (a). How is the acidic nature suspected?
- The two gases A and B are bleaching agents. A is greenish-yellow and bleaches due to its oxidizing property while B is a colourless gas that bleaches due to reduction. Identify A and B?
- Which gas turn blue cobalt chloride paper light pink?
Give one similarity in the test between- Cl2 and HCl
- SO2 and CO2.
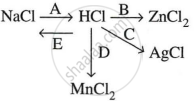
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
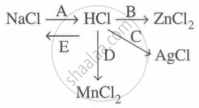
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.