Advertisements
Advertisements
Question
Give a balanced equation when dilute hydrochloric acid is added to : Sodium sulphite
Solution
Na2SO3 + 2HCl → 2NaCl + SO2 +H2O
APPEARS IN
RELATED QUESTIONS
Give reason for the following:
silver nitrate solution can be used to distinguish HCl from HNO3
Complete the following reaction and balance them.
Zn+ 2HCl ⟶
Distinguish between the following pairs of compounds using the reagent given in the bracket.
Mangenese dioxide and copper (II) oxide. (using concentrated HCl)
Name the following :
Acid present in the stomach of mammals.
How will you prove that hydrochloric acid contains
- hydrogen
- chlorine?
Write equations for the reactions.
Complete and balance the following reaction, state whether dilutes or conc. acid is used.
\[\ce{NH4OH + HCl -> }\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH +HCI ->}\]
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl->}\]
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
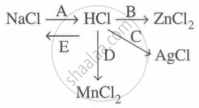
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.

