Advertisements
Advertisements
Question
How will you prove that hydrochloric acid contains
- hydrogen
- chlorine?
Write equations for the reactions.
Solution
We can prove that hydrochloric acid contains both hydrogen and chlorine by the following experiment:
Take a voltameter used for water electrolysis with a platinum cathode and a graphite anode. Pour 4 molars of \[\ce{HCl}\] into the voltameter and measure the direct electric current. The cathode emits a colorless gas, while the anode produces a greenish-yellow gas. If you put a burned splinter close to a colorless gas, the gas will burst into flames, showing that it is hydrogen gas. It is clear that the gas is chlorine because when wet starch iodide paper is put in it, it turns blue-black.
Chemical equation: \[\ce{2HCl -> H2 ^ + Cl2 ^}\]
This experiment proves that hydrochloric acid contains both hydrogen and chlorine.
APPEARS IN
RELATED QUESTIONS
Name a black metallic oxide which reacts with hydrochloric acid to give a coloured solution.
Give reason for the following:
silver nitrate solution can be used to distinguish HCl from HNO3
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl -> \underline{\phantom{..........}}}\].
Give a balanced equation when dilute hydrochloric acid is added to : Sodium sulphite
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl->}\]
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl ->}\]
Convert hydrochloric acid to nascent chlorine.
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
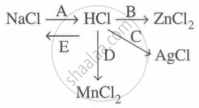
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.