Advertisements
Advertisements
प्रश्न
How will you prove that hydrochloric acid contains
- hydrogen
- chlorine?
Write equations for the reactions.
उत्तर
We can prove that hydrochloric acid contains both hydrogen and chlorine by the following experiment:
Take a voltameter used for water electrolysis with a platinum cathode and a graphite anode. Pour 4 molars of \[\ce{HCl}\] into the voltameter and measure the direct electric current. The cathode emits a colorless gas, while the anode produces a greenish-yellow gas. If you put a burned splinter close to a colorless gas, the gas will burst into flames, showing that it is hydrogen gas. It is clear that the gas is chlorine because when wet starch iodide paper is put in it, it turns blue-black.
Chemical equation: \[\ce{2HCl -> H2 ^ + Cl2 ^}\]
This experiment proves that hydrochloric acid contains both hydrogen and chlorine.
APPEARS IN
संबंधित प्रश्न
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl -> \underline{\phantom{..........}}}\].
Complete the following reaction and balance them.
Ca(HCO3)2 + 2HCl →
Name the following:
Acid used to extract glue from bones.
Calcium oxide and phosphorous pentoxide are very good drying agents but they are not used to dry HCI gas. Give reasons for each.
Give a balanced equation when dilute hydrochloric acid is added to : Zinc Metal
How will you identify?
Chloride ion
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution:
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | _________ | _________ |
| 2. | Magnesium ribbon | _________ | _________ |
| 3. | Manganese (IV) oxide with heating | _________ | _________ |
| 4. | Sodium sulphide | _________ | _________ |
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
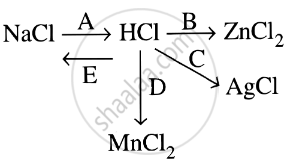
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH +HCI ->}\]
Convert Hydrochloric acid to nascent chlorine.
