Advertisements
Advertisements
प्रश्न
How will you identify?
Chloride ion
उत्तर
A small amount of the salt is taken in a test tube and conc. H2SO4 is added to it and then test tube is warmed, if a colourless gas with pungent odour is evolved then chloride ions are present in the salt. It can be confirmed by bringing a glass rod dipped in ammonia solution near the gas evolved, if dense white fumes are formed then presence of chloride ions is confirmed.
APPEARS IN
संबंधित प्रश्न
Name the experiment which demonstrates that hydrogen chloride is soluble in water.
Give reason for the following:
silver nitrate solution can be used to distinguish HCl from HNO3
Name the gas evolved when dilute hydrochloric acid is added to: Magnesium bicarbonate
Give a balanced equation when dilute hydrochloric acid is added to : Calcium carbonate
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl->}\]
Write an equation for the reaction of hydrochloric acid on:
Manganese (IV) oxide
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NA4OH + HCl -> }\]
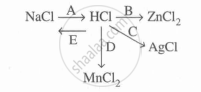
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
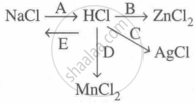
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
NH4OH + HCl ⟶
