Advertisements
Advertisements
Question
Write the structures of optical isomers of the complex ion `[Co(en)_2Cl_2]^+`
Solution
`[Co(en)_2Cl_2]^+`
C is form
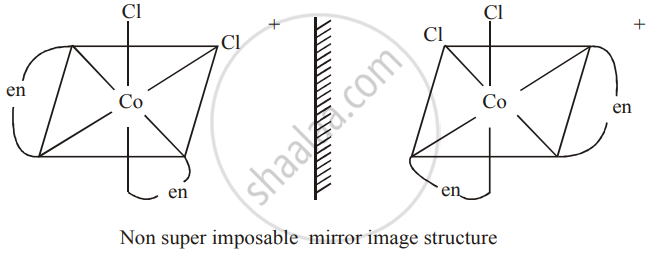
APPEARS IN
RELATED QUESTIONS
Define the following term:
Anomers
How many geometrical isomers are possible in the following coordination entity?
[Cr(C2O4)3]3−
Draw the structure of optical isomers of [Cr(NH3)2Cl2(en)]+.
Draw all the isomers (geometrical and optical) of [Co(NH3)2Cl2(en)]+.
Write all the geometrical isomers of [Pt(NH3)(Br)(Cl)(py)] and how many of these will exhibit optical isomers?
Name the type of isomerism shown by the following pair of compounds:
[CoCl(H2O)(NH3)4]Cl2 and [CoCl2(NH3)4]Cl.H2O
Write the IUPAC name of [Co(en)2Cl2]+ ion and draw the structures of its geometrical isomers.
Indicate the types of isomerism exhibited by the following complex and draw the structure for these isomer: \[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the types of isomerism exhibited by the following complexes and draw the structure for these isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
