Advertisements
Advertisements
Question
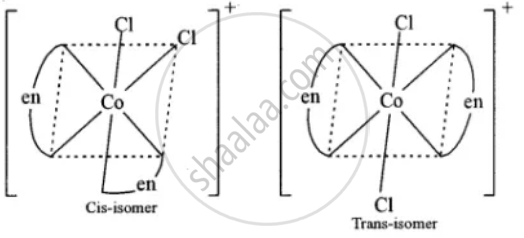
Write the IUPAC name of [Co(en)2Cl2]+ ion and draw the structures of its geometrical isomers.
Solution
dichloro bis (ethylenediamine) cobalt (III) ion.
APPEARS IN
RELATED QUESTIONS
When a coordination compound CoCl3.6NH3 is mixed with AgNO3, 3moles of AgCl are precipitated per mole of the compound. Write
(i) Structural formula of the complex
Draw the geometrical isomers of complex \[\ce{[Co(en)2Cl2]+}\].
How many geometrical isomers are possible in the following coordination entity?
[Co(NH3)3Cl3]
Draw the structure of optical isomers of [PtCl2(en)2]2+.
Draw the structure of optical isomers of [Cr(NH3)2Cl2(en)]+.
Write all the geometrical isomers of [Pt(NH3)(Br)(Cl)(py)] and how many of these will exhibit optical isomers?
What type of isomers are `[Co(NH_3)_5Br]SO_4`and `[Co(NH_3)_5SO_4]Br`?. Give a chemical test to distinguish between them.
What type of structural isomers are [Co(NH3)5 Br] SO4 and [Co(NH3)5 SO4]Br? Give a chemical test to distinguish the isomers.
Which of the following molecules has a chiral centre correctly labelled with an asterisk (*)?
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
