Advertisements
Advertisements
Question
Write the Lewis structure of hydrogen peroxide.
Short Note
Solution
\[\ce{H2O2}\] structure in gas phase, dihedral angle is 111.50. \[\ce{H2O2}\] structure in solid phase at 110 k, dihedral angle is 90.20.
(a) Gas-phase
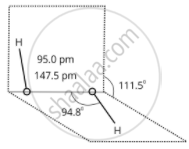
(b) Solid-phase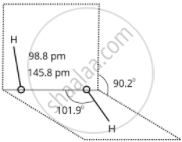
shaalaa.com
Structure of Hydrogen Peroxide
Is there an error in this question or solution?
APPEARS IN
RELATED QUESTIONS
Draw the gas phase and solid phase structure of \[\ce{H2O2}\].
How will you concentrate \[\ce{H2O2}\]? Show differences between structures of \[\ce{H2O2}\] and \[\ce{H2O}\] by drawing their spatial structures. Also mention three important uses of \[\ce{H2O2}\].
A colourless liquid ‘A’ contains H and O elements only. It decomposes slowly on exposure to light. It is stabilised by mixing urea to store in the presence of light. Suggest possible structure of A.
