Advertisements
Advertisements
Question
How will you concentrate \[\ce{H2O2}\]? Show differences between structures of \[\ce{H2O2}\] and \[\ce{H2O}\] by drawing their spatial structures. Also mention three important uses of \[\ce{H2O2}\].
Solution
Hydrogen peroxide is produced by acidifying barium peroxide and eliminating surplus water by evaporation under low pressure. Water is used to extract it, then distillation under lower pressure concentrates it to around 30% (by mass). Careful distillation under low pressure can increase the concentration to 85%. To achieve pure \[\ce{H2O2}\], the residual water can be frozen out.
Spatial structures of \[\ce{H2O2}\] and \[\ce{H2O}\]:
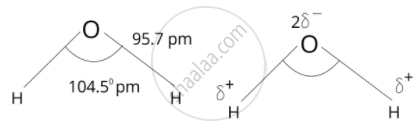
Uses of \[\ce{H2O2}\]:
(i) As an antiseptic it is sold in the market as perhydrol.
(ii) It is used to manufacture chemicals like sodium perborate and per- carbonate. It is employed in the industries as a bleaching agent for textiles.
(iii) It is used in laboratories as an oxidising agent. Also used in rocket fuels.
APPEARS IN
RELATED QUESTIONS
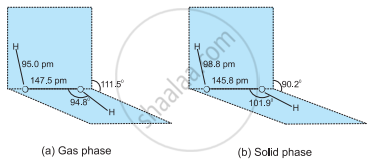
Draw the gas phase and solid phase structure of \[\ce{H2O2}\].
Write the Lewis structure of hydrogen peroxide.
A colourless liquid ‘A’ contains H and O elements only. It decomposes slowly on exposure to light. It is stabilised by mixing urea to store in the presence of light. Suggest possible structure of A.
