English Medium
Academic Year: 2021-2022
Date & Time: 10th May 2022, 10:30 am
Duration: 2h
Advertisements
General Instructions :
Read the following instructions carefully and strictly follow them:
-
This Question paper contains 15 questions. All questions are compulsory.
-
This question paper is divided into three Sections viz. Sections A, B, and C.
-
Section-A—Question number 1 to 7 are short answer type questions. Each question carries two marks.
-
Section-B—Question number 8 to 13 are also short answer type questions. Each question carries three marks.
-
Section-C—Question number 14 to 15 are case-based questions. Each question carries four marks.
-
Internal choices have been provided in some questions. Only one of the alternatives has to be attempted.
Explain giving reason why although the nuclear charge in atoms increases in moving from left to right in a period as well as in moving from top to bottom in a group in the Modern periodic table, but the size of the atoms does not vary similarly in both situations.
Chapter: [0.05] Periodic Classification of Elements
As shown in the diagram an aluminium rod 'AB' is suspended horizontally between the two poles of a strong horse shoe magnet in such a way that the axis of rod is horizontal and the direction of the magnetic field is vertically upward. The rod is connected in series with a battery and a key.
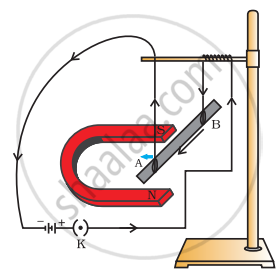
State giving reason:
- What is observed when a current is passed through the aluminium rod from end B to end A?
- What change is observed in a situation in which the axis of the rod 'AB' is moved and aligned parallel to the magnetic field and current is passed in the rod in the same direction?
Chapter: [0.12] Magnetic Effects of Electric Current
"Magnetic field is a physical quantity that has both direction and magnitude." How can this statement be proved with the help of magnetic field lines of a bar magnet?
Chapter: [0.12] Magnetic Effects of Electric Current
Consider the following molecular formulae of carbon compounds:
(i) CH3COOH (ii) CH3OH (iii) C2H6 (iv) C3H4 (v) C4H8
- Which one of these compounds belongs to homologous series of alcohols?
- Identify the compound having triple bond between carbon-carbon atoms.
- Write the molecular formula of the first member of the homologous series to which CH3COOH belongs.
- Write the general formula of the series to which the compound C4H8 belongs.
Chapter: [0.04] Carbon and its Compounds
Describe the function of placenta.
Chapter: [0.07] How do Organisms Reproduce?
State the function of Fallopian tubes.
Chapter: [0.07] How do Organisms Reproduce?
Mention the functions of Uterus in the human female reproductive system.
Chapter: [0.07] How do Organisms Reproduce?
Advertisements
Mention the functions of Ovary.
Chapter: [0.07] How do Organisms Reproduce?
What is Regeneration?
Chapter: [0.07] How do Organisms Reproduce?
Explain the process of regeneration in Planaria.
Chapter: [0.07] How do Organisms Reproduce?
Differentiate between binary fission in Amoeba and binary fission in Leishmania.
Chapter: [0.07] How do Organisms Reproduce?
How does reproduction take place in malarial parasites?
Chapter: [0.07] How do Organisms Reproduce?
State the modern periodic law.
Chapter: [0.05] Periodic Classification of Elements
How many rows and columns are there in the Modem Periodic Table?
Chapter: [0.05] Periodic Classification of Elements
Name the elements whose compounds formed the basis of classification in Mendeleev's periodic table. Why did Mendeleev choose these elements?
How the formulae of these compounds had helped Mendeleev in deciding the position of an element in his periodic table?
Chapter: [0.05] Periodic Classification of Elements
Why are autotrophs considered to be at the first trophic level of all food chains?
Chapter: [0.13] Our Environment
State the reason for limited number of trophic levels in nature.
Chapter: [0.13] Our Environment
In flowering plants, the pollen grains are transferred to stigma by pollination but the female germ cells are present in the ovary.
Explain with the help of a labelled diagram (only concerned parts), how the male germ cell reaches the ovary.
Chapter: [0.07] How do Organisms Reproduce?
Advertisements
"Two different forms of carbon - diamond and graphite have different structure and very different physical properties even though their chemical properties are same." Explain why.
Chapter: [0.04] Carbon and its Compounds
State the reasons, why carbon cannot
- Lose four electrons to form C4+ cation and
- Gain four electrons to form C4- anion.
How does carbon overcome this problem to form compounds?
Chapter: [0.03] Metals and Non Metals [0.04] Carbon and its Compounds
A student wants to use an electric heater, an electric bulb and an electric fan simultaneously.
How should these gadgets be connected with the mains? Justify your answer giving three reasons.
Chapter: [0.11] Electricity
What is an electric fuse?
Chapter: [0.12] Magnetic Effects of Electric Current
How is electric fuse connected in a circuit?
Chapter: [0.12] Magnetic Effects of Electric Current
Draw a diagram showing flow of energy from solar input to a food chain having four trophic levels in an ecosystem. List two inference that can be drawn from the energy flow diagram.
Chapter: [0.13] Our Environment
AB is a coil of copper wire having a large number of turns. The ends of the coil are connected with a galvanometer as shown. When the north pole of a strong bar magnet is moved towards end B of the coil, a deflection is observed in the galvanometer.

- State the reason for using galvanometer in the activity and why does its needle deflects momentarily when magnet is moved towards the coil.
- What would be observed in the galvanometer in a situation when the coil and the bar magnet both move with the same speed in the same direction? Justify your answer.
- State the conclusion that can be drawn from this activity.
Will there be any change in the momentary deflection in the galvanometer if number of turns in the coil is increased and a more stronger magnet is moved towards the coil?
OR
What is electromagnetic induction? What is observed in the galvanometer when a strong bar magnet is held stationary near one end of a coil of large number of turns? Justify your answer.
Chapter: [0.12] Magnetic Effects of Electric Current
Sex of an individual is determined by different factors in various species. Some animals rely entirely on the environmental ones, while in some other animals the individuals can change their sex during their life time indicating that sex of some species is not genetically determined. However in human beings, the sex of an individual is largely determined genetically.
- In what way are the sex chromosomes 'X' and 'Y' different in size? Name the mismatched pair of sex chromosome in humans.
- Write the number of pair/pairs of sex chromosomes present in human beings. In which one of the parent (male/female) perfect pair/pairs of sex chromosome are present?
- Citing two examples, justify the statement "Sex of an individual is not always determined genetically".
OR
Draw a flow chart to show that sex is determined genetically in human beings.
Chapter: [0.08] Heredity
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 10 Science with solutions 2021 - 2022
Previous year Question paper for CBSE Class 10 Science-2022 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 10.
How CBSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Science will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
