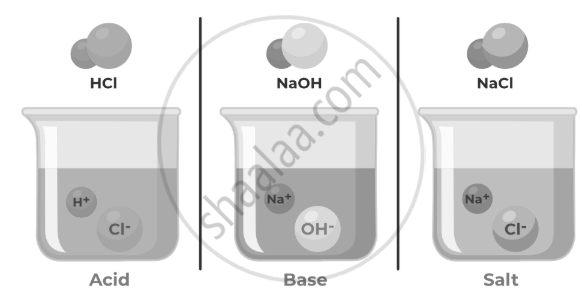
Acids are chemical substances that release hydrogen ions (H⁺) when dissolved in water. They are found naturally in foods like citrus fruits and vinegar and are widely used in industries.
Classification:
- Natural (Organic Acids): Found in nature, e.g., acetic acid (vinegar) and citric acid (fruits).
- Mineral Acids (Inorganic Acids): Synthetic or derived from minerals, e.g., hydrochloric acid (HCl), sulphuric acid (H₂SO₄).
Properties:
- Sour taste, with a burning or tingling sensation.
- React with bases to form salt and water (neutralisation).
- pH less than 7.
Applications: Used in industries for fertilisers, dyes, cleaning, and manufacturing.
Types of Acids:
1. Based on Occurrence
- Natural Acids: Derived from plants or animals, e.g., acetic acid (CH₃COOH).
- Mineral Acids: Man-made or extracted from minerals, e.g., hydrochloric acid (HCl).
2. Based on Concentration:
- Strong Acids: Fully ionise in water, e.g., HCl, H₂SO₄.
- Weak Acids: Partially ionise in water, e.g., acetic acid (CH₃COOH).
Acids are essential in both natural and industrial processes, making them a crucial part of chemical applications.