Topics
The Language of Chemistry
- Matter (Substance)
- Molecules
- Pure Substances
- Elements
- Classification of Molecules
- Symbols Used to Represent Atoms of Different Elements
- Valency
- Variable Valency
- Chemical Formula or Molecular Formula
- Ions (Radicals) and Its Types
- Chemical Formula or Molecular Formula
- Naming Chemical Compounds
- To Calculate the Valency from the Formula
- Chemical Equation
- Balancing Chemical Equation
- Atomic Mass
- Molecular Mass
- Percentage Composition, Empirical and Molecular Formula
- Empirical Formula of a Compound
- Hydrogen
Chemical Changes and Reactions
Water
- Water: Our Lifeline
- Physical Properties of Water
- Chemical Properties of Water
- Water - a Universal Solvent
- Solutions as 'Mixtures' of Solids in Water
- Components of Solutions
- Different Types of Solutions
- Saturated Solutions
- Concentration of a Solution
- Solubility
- Crystals and Crystallisation
- Hydrated and Anhydrous Substances
- Efflorescence, Hygroscopic, and Deliquescence Substances
- Drying and Dehydrous Substances
- Classification of water: Soft and Hard Water
- Causes of Hardness
- Advantage and Disadvantage of Hard Water
- Removal of Hardness of Water
- Salts
- Prevention of Water Pollution
Atomic Structure and Chemical Bonding
- Chemical Bond
- History of Atom
- Dalton’s Atomic Theory
- Elements
- Atoms: Building Blocks of Matter
- Discovery of Charged Particles in Matter
- Electrons (e)
- Protons (p)
- Nucleus
- Neutrons (n)
- J. J. Thomson’s Atomic Model
- Lord Rutherford’s Atomic model
- Neils Bohr’s Model of an Atom
- Structure of an Atom
- Atomic Number (Z), Mass Number (A), and Number of Neutrons (n)
- Atomic Mass
- Electronic Configuration of Atom
- Valency
- Reason for Chemical Activity of an Atom
- Isotopes
- Isobars
- Ionic or Electrovalent Bond
- Ionic or Electrovalent Bond
- The Covalent Bond
- Types of Covalent Bond
- Formation of Covalent Bond
The Periodic Table
- History of Periodic Table: Early Attempts at the Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Merits and Demerits of Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Advantage and Disadvantage of Modern Periodic Table
- Periodic Properties
- Shells (Orbits)
- Valency
- Properties of Elements
- Atomic Radius Or Atomic Size
- Metallic and Non-metallic Characters
- Study of Specific Groups in Periodic Table
- Group I (Alkali Metals)
- Group II (Alkaline Earth Metals)
- Group VIIA Or Group 17 (The Halogens)
- Group Zero or 18 Group (Noble Gases)
- Uses of Periodic Table
- Types of Element: Metals
Study of the First Element - Hydrogen
- Position of Hydrogen in Periodic Table
- Similarities Between Hydrogen and Alkali Metals
- Similarities Between Hydrogen and Halogens
- Hydrogen
- Preparation of Hydrogen
- Application of Activity Series in the Preparation of Hydrogen
- Laboratory Preparation of Hydrogen
- Manufacture of Hydrogen
- Physical Properties of Hydrogen
- Chemical Properties of Hydrogen
- Uses of Hydrogen
- Oxidation, Reduction and Redox Reactions
Study of Gas Laws
- Gases and Its Characteristics
- Molecular Motion : Relationship of Temperature, Pressure and Volume
- The Gas Laws
- Pressure and Volume Relationship or Bolye's Law
- Temperature - Volume Relationship or Charles's Law
- Absolute Zero
- The Temperature and a Thermometer
- Scales of Thermometers
- Gas Equation
- Standard Temperature Pressure (S.T.P.)
- The Effect of Moisture and Pressure
Atmospheric Pollution
- Atmospheric Pollution
- Air Pollution and Its Causes
- Effects of Air Pollution
- Prevention of Air Pollution
- Gaseous Pollutants and Their Effects
- Acid Rain
- Causes of Acid Rain
- Green House Effect
- Advantage of Green House Effect
- Global Warming
- Preventive Measures of Global Warming
- Ozone
- Ozone Layer Depletion
Elements, Compounds and Mixtures
- Differences Between Elements, Compounds, and Mixtures
- Types of Mixtures
- Mixture
- Separation of Mixtures
- Use of Solvent and Filtration
- Concept of Evaporation
- Simple Distillation Method
- Simple Distillation Method
- Chromatography Method
- Centrifugation Method
- Solvent Extraction (Using a Separating Funnel Method)
Matter and Its Composition: Law of Conservation of Mass
- Heat and change of physical state
- Inter-particle Space and Interparticle Attraction and Collision
- Law of Conservation of Mass
Practical Work
- Laboratory Preparation of Hydrogen
- Laboratory Preparation of Oxygen
- Laboratory Preparation of Carbon Dioxide
- Laboratory Preparation of Chlorine
- Laboratory Preparation of Hydrogen Chloride Gas
- Laboratory Preparation of Sulphur Dioxide
- Laboratory Preparation of Hydrogen Sulphide
- Laboratory Preparation of Ammonia Gas
- Laboratory Preparation of Water Vapour
- Laboratory Preparation of Nitrogen Dioxide
- Action of Heat on a Given Substance
- Action of Dilute Sulphuric Acid on a Given Substance
- Dry Test
- Recognition of Substances by Colour
- Recognition of Substances by Odour
- Recognition of Substances by Physical State
- Recognition of Substances by Action of Heat
- Flame Test
- Classification of water: Soft and Hard Water
- Simple Experiments Based on Hard Water and Soft Water
- Water Pollution and Its Causes
- Water Quality
- Strength of Acidic or Basic Solutions
- Prevention of Water Pollution
- Distillation Method
- Experiment 1
- Experiment 2
Distillation Method:
Distillation is a process used to separate components of a liquid mixture based on their different boiling points. It involves heating a liquid to form vapour and condensing the vapour back into liquid form. This method is commonly used to separate water from salt solutions and for the purification of impure liquids.
Experiment 1
1. Aim: To separate a mixture of acetone and water using the distillation method.
2. Requirements: Distillation flask, a mixture of acetone and water, thermometer, condenser, etc.
3. Principle: Acetone has a lower boiling point than water, so it vaporises faster. The temperature should not exceed the boiling point of water, which is determined with the help of a thermometer. Acetone will turn into liquid when it passes through the condenser and water will be left behind in the distillation flask. Hence, pure acetone can be obtained.

4. Procedure
- Take the mixture in a distillation flask.
- Fit it with a thermometer and arrange the apparatus as shown
- Heat the mixture slowly, keeping a close watch on the thermometer.
- The acetone vaporises and condenses in the condenser and can be collected from the condenser outlet.
5. Observation: Acetone got vaporised, and while passing through the condenser, it got converted into liquid form again.
6. Inference/Result: Acetone is a volatile liquid and vaporises easily on heating, and water remains in the flask until its boiling point is not received. On cooling, acetone again converts into its liquid form, which is free of water.
Experiment 2
1. Aim: To separate pure water from a salt solution through distillation.
2. Requirements: round-bottom flask, conical flask, saltwater, wire gauze, burner, condenser (tube for condensation), rubber tubing for cooling water supply, tripod stand.
3. Procedure
- Place salt water in the round-bottom flask. Connect the flask to a condenser tube (cooled by circulating water).
- Place the conical flask at the end of the condenser to collect the distilled water.
- Heat the salt water using a burner, with the flask on a wire gauze for stability.
4. Observation
- As the water boils, it forms steam. The steam passes through the condenser, which cools and condenses into water droplets.
- These droplets collect in the conical flask. Once all the water is collected, salt remains in the round-bottom flask.
5. Conclusion: The salt stays behind, and the pure water is collected in the conical flask. Distillation separates soluble substances (like salt) from liquids by evaporating and condensing the liquid. It can also be used to purify impure liquids.
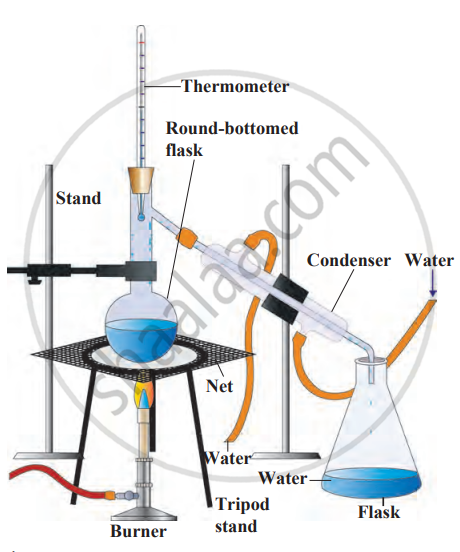
Distillation method
