Advertisements
Advertisements
प्रश्न
10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount of HCl solution (the same solution as before) required to neutralise it will be:
विकल्प
4 mL
8 mL
12 mL
16 mL
उत्तर
16 mL
Explanation:
Since 10 mL of NaOH requires 8 mL of HCl, 20 mL of NaOH requires 8 × 2 = 16 mL of HCl.
संबंधित प्रश्न
Dilute NaOH can be tested with
(a) red litmus paper
(b) blue litmus paper
(c) lime water
(d) Na2CO3
How is the concentration of hydroxide ions (OH−) affected when excess base is dissolved in a solution of sodium hydroxide?
The litmus paper or the litmus solution is obtained from ________ plant
(A) Moss
(B) Lichen
(C) Rose
(D) Hibiscus
Complete and balance the following chemical equations:
CuO (s) + HCI (aq) →
What happens when an acid reacts with a metal oxide? Explain with the help of an example. Write a balanced equation for the reaction involved.
Compounds such as alcohol and glucose also contain hydrogen but are not categorised as acids. Describe an activity to prove it.
Write a balanced chemical equation of the reaction which takes place.
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
On the basis of the above reaction, what can you say about the nature of copper oxide?
Wasp stings can be treated with:
(a) baking soda
(b) vinegar
(c) washing soda
(d) milk of magnesia
What happens when a solution of sodium hydrogencarbonate is heated? Write equation of the reaction involved.
Name a metal compound which has detergent properties (cleansing properties).
Choose the correct alternative and rewrite the following sentence.
Phenolphthalein is ___________ type of indicator.
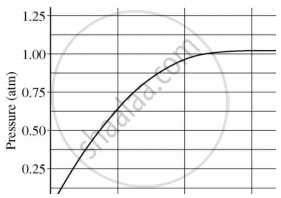
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
During the preparation of hydrogen chloride gas on a humid day, the gas is usually passed through the guard tube containing calcium chloride. The role of calcium chloride taken in the guard tube is to ______
Which of the following salts does not contain any water of crystallisation?
What happens when nitric acid is added to egg shell?
What are strong and weak acids? In the following list of acids, separate strong acids from weak acids.
Hydrochloric acid, citric acid, acetic acid, nitric acid, formic acid, sulphuric acid.
Acids present in fruits and vegetables are called ______ acids.
Take a clean test tube with a holder and pour some dilute hydrochloric acid. Add a few pieces of magnesium ribbon pieces slowly. What do you observe? Now show a burning match stick near the mouth of the test tube. Do you hear any sound? The gas burns with a pop sound. From this, it is observed that hydrogen gas has been formed due to the reaction between acid and metal.
Give a balanced equation for the reaction:
Silver nitrate solution and sodium chloride solution.
