Advertisements
Advertisements
प्रश्न
A 50 kg man is running at a speed of 18 km h−1. If all the kinetic energy of the man can be used to increase the temperature of water from 20°C to 30°C, how much water can be heated with this energy?
उत्तर
Given:-
Mass of the man, m = 50 kg
Speed of the man, v = 18 km/h = `18xx5/18=5"m/s"`
Kinetic energy of the man is given by
`K=1/2mV^2`
`K=(1/2)50xx5^2`
`K=25xx25=625 J`
Specific heat of the water, s = 4200 J/Kg-K
Let the mass of the water heated be M.
The amount of heat required to raise the temperature of water from 20°C to 30°C is given by
Q = msΔT = M × 4200 × (30 − 20)
Q = 42000 M
According to the question,
Q = K
42000 M = 625
`rArrM=625/42xx10^-3`
`=14.88xx10^-3`
=15 kg
APPEARS IN
संबंधित प्रश्न
Explain the following:
How can you relate the formation of water droplets on the outer surface of a bottle taken out of refrigerator with formation of dew?
1 kg of ice at 0°C is mixed with 1 kg of steam at 100°C. What will be the composition of the system when thermal equilibrium is reached? Latent heat of fusion of ice = 3.36 × 103 J kg−1 and latent heat of vaporization of water = 2.26 × 106 J kg−1.
A calorimeter contains 50 g of water at 50°C. The temperature falls to 45°C in 10 minutes. When the calorimeter contains 100 g of water at 50°C, it takes 18 minutes for the temperature to become 45°C. Find the water equivalent of the calorimeter.
Explain the following
A hollow glass sphere which floats with its entire volume submerged in water at 4°C, sinks when water is heated above 4°C.
A deep pond of water has its top layer frozen. What will be the likely temperature of water layer at the bottom of the pond?
What are land and sea breezes? Explain with the help of a labeled diagram.
In cold countries, the water pipes are covered with poor conductors. Why?
Why does a thick glass tumbler crack when very hot water is poured in it?
What is the criterion of choosing the two metals for a bimetal strip?
Observe the given graph and answer the following questions:
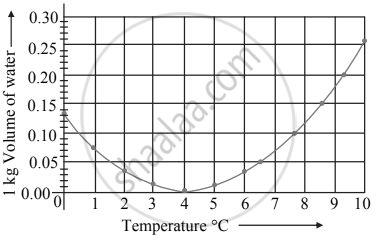
- Name the process represented in the figure.
- At what temperature does this process take place?
In cold regions, in winter the pipes for water supply break.
Observe the following diagram and write the answers to the questions given below.
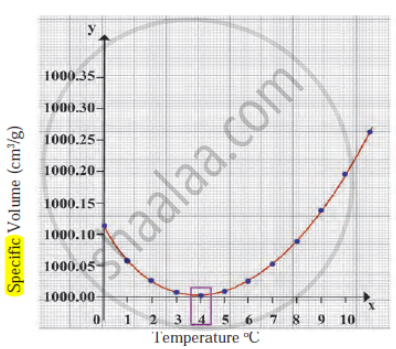
- Which process does the graph represent?
- What is the range of temperature responsible for the behaviour?
Name the following diagram appropriately.

Observe the given picture and answer the following questions.
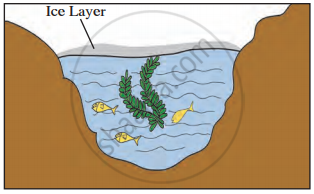
- Which property do you understand in this picture?
- What is the temperature of the water at the surface?
- What is the temperature below the layer of ice on the surface?
A graph between the volume and temperature of water is shown. Explain the anomalous behaviour of water.

