Advertisements
Advertisements
Question
A 50 kg man is running at a speed of 18 km h−1. If all the kinetic energy of the man can be used to increase the temperature of water from 20°C to 30°C, how much water can be heated with this energy?
Solution
Given:-
Mass of the man, m = 50 kg
Speed of the man, v = 18 km/h = `18xx5/18=5"m/s"`
Kinetic energy of the man is given by
`K=1/2mV^2`
`K=(1/2)50xx5^2`
`K=25xx25=625 J`
Specific heat of the water, s = 4200 J/Kg-K
Let the mass of the water heated be M.
The amount of heat required to raise the temperature of water from 20°C to 30°C is given by
Q = msΔT = M × 4200 × (30 − 20)
Q = 42000 M
According to the question,
Q = K
42000 M = 625
`rArrM=625/42xx10^-3`
`=14.88xx10^-3`
=15 kg
APPEARS IN
RELATED QUESTIONS
How does the anomalous expansion of water help aquatic organisms in cold climates?
A calorimeter contains 50 g of water at 50°C. The temperature falls to 45°C in 10 minutes. When the calorimeter contains 100 g of water at 50°C, it takes 18 minutes for the temperature to become 45°C. Find the water equivalent of the calorimeter.
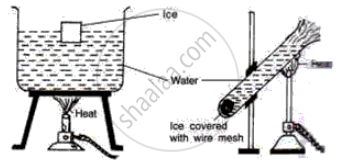
Describe an experiment to show that water has maximum density at 4°C. What important consequences follow this peculiar property of water? Discuss the importance of this phenomenon in nature.
Explain the following
A glass bottle completely filled with water and tightly closed at room temperature is likely to burst when kept in the freezer of a refrigerator.
Explain why do vegetables and fruits get damaged during severe frost?
In cold countries, the water pipes are covered with poor conductors. Why?
The following figure shows a metal cylinder, containing boiling water. One half side A is polished and another half, B is painted black. Two thin metal sheets X and Y are painted black and have one rubber stopper fixed with wax on each sheet. These sheets are equidistant from the boiling water (container A, B) as shown in the diagram. What would you expect to happen after a few minutes? Give a reason for your answer.
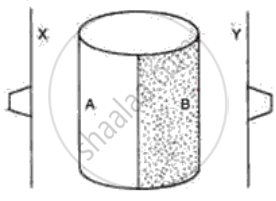
Study the following diagrams and write down your observations.
What will be the approximate temperature of water in the lake shown in the following diagram?
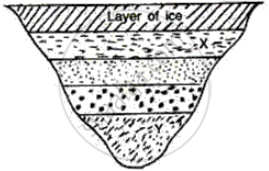
(i) At X, and
(ii) At Y?
Observe the given graph and answer the following questions:
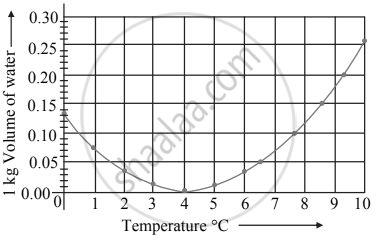
- Name the process represented in the figure.
- At what temperature does this process take place?
When the temperature of water decreases below 4 °C it’s volume _______.
In a region with a cold climate the aquatic animals can survive at 4 °C, because _______.
Observe the following diagram and write the answers to the questions given below.
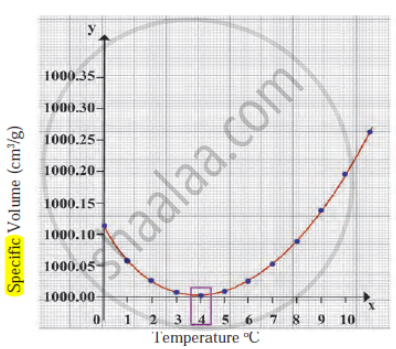
- Which process does the graph represent?
- What is the range of temperature responsible for the behaviour?
Name the following diagram appropriately.

Write a short note.
Anomalous behaviour of water
