Advertisements
Advertisements
Question
What will be the approximate temperature of water in the lake shown in the following diagram?
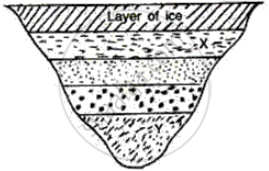
(i) At X, and
(ii) At Y?
Solution
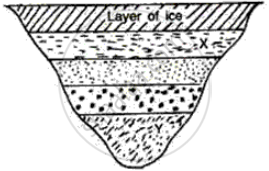
The layer at the top level is ice so its temperature is therefore 0°C and temperature at every layer of water below ice increases by 1°C
(i) At X = 1°C since it is the first layer below the ice
(ii) At Y = 4°C since it is the fourth layer below ice
APPEARS IN
RELATED QUESTIONS
How does the anomalous expansion of water help aquatic organisms in cold climates?
Observe the following graph. Considering the change in volume of water as its temperature is raised from 0°C, discuss the difference in the behaviour of water and other substances. What is this behaviour of water called?

Calculate the time required to heat 20 kg of water from 10°C to 35°C using an immersion heater rated 1000 W. Assume that 80% of the power input is used to heat the water. Specific heat capacity of water = 42000 J kg−1 K−1.
Describe an experiment to show that water has maximum density at 4°C. What important consequences follow this peculiar property of water? Discuss the importance of this phenomenon in nature.
Explain, why are the exposed water pipes lagged with straw during severe winter?
What are land and sea breezes? Explain with the help of a labeled diagram.
What is the criterion of choosing the two metals for a bimetal strip?
Write scientific reason.
Placing a plastic bottle filled with water in the freezing compartment in the freezer can cause the bottle to explode.
Match the columns:
| Column ‘A’ | Column ‘B’ |
| The density of water is maximum at | (a) 0°C |
| (b) 4°C | |
| (c) 100°C |
