Advertisements
Advertisements
Question
The diagram shows a sketch graph showing a change in volume of a given mass of ice at -5°C to water at 15°C.
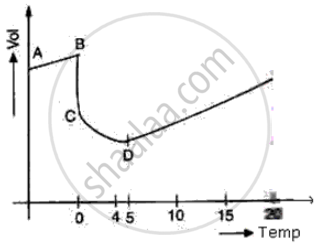
(i) Why does the temperature along BC remain constant?
(ii) When is the volume of water minimum?
(iii) At what temperature does water have maximum density? Explain.
Solution
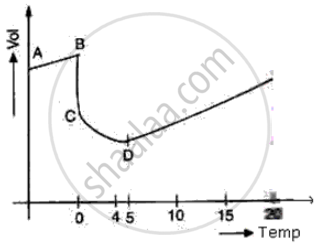
(i) At BC temperature is constant because at 0°C all of the heat given is used in the process of formation of water from ice and no part of the heat is used in changing the temperature of ice.
(ii) At point D i.e. at 4°C the volume of water is minimum
(iii) At point D i.e. at 4°C, the density of water is maximum due to anomalous expansion of water in the temperature range of 0°C and 4°C as volume decreases, the density of water increases
APPEARS IN
RELATED QUESTIONS
What is matter? What is it composed of?
What do you understand by thermal expansion of a substance?
100 ml of each of the following liquid is heated from 10°C to 50°C. Which will expand more : (a) water (b) benzene (c) alcohol?
An iron washer is heated. State the effect on its external.
Give reasons for the following:
Telephone wires sag in summer.
Solids ______ on heating and ______ on cooling.
How solids are expanded on heating?
When we cool the liquid it will become ______.
