Advertisements
Advertisements
प्रश्न
The diagram shows a sketch graph showing a change in volume of a given mass of ice at -5°C to water at 15°C.
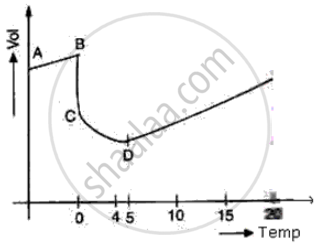
(i) Why does the temperature along BC remain constant?
(ii) When is the volume of water minimum?
(iii) At what temperature does water have maximum density? Explain.
उत्तर
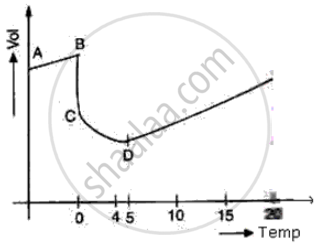
(i) At BC temperature is constant because at 0°C all of the heat given is used in the process of formation of water from ice and no part of the heat is used in changing the temperature of ice.
(ii) At point D i.e. at 4°C the volume of water is minimum
(iii) At point D i.e. at 4°C, the density of water is maximum due to anomalous expansion of water in the temperature range of 0°C and 4°C as volume decreases, the density of water increases
APPEARS IN
संबंधित प्रश्न
Equal volumes of different liquids expand by a different amount when they are heated to the same rise in temperature.
What is matter? What is it composed of?
Two iron rods – one 10 m long and the other 5 m long, are heated to the same rise in temperature. Which will expand more?
Iron rims are heated before they are fixed on the wooden wheels.
Why is one end of a steel girder in a bridge kept on rollers instead of fixing it in pillar?
An iron washer is heated State the effect on its density.
Complete the following sentence:
When ice melts its volume ______ and density _______.
Explain thermal expansion with suitable examples.
- Assertion: Gas is highly compressible than solid and liquid
- Reason: Interatomic or intermolecular distance in the gas is comparably high.
Analogy:
Expansion is length:: Linear expansion.
Expansion in volume:: ______.
