Advertisements
Advertisements
प्रश्न
What will be the approximate temperature of water in the lake shown in the following diagram?
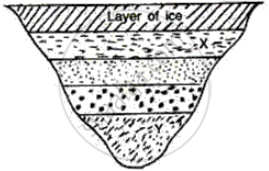
(i) At X, and
(ii) At Y?
उत्तर
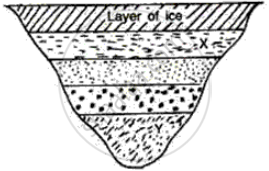
The layer at the top level is ice so its temperature is therefore 0°C and temperature at every layer of water below ice increases by 1°C
(i) At X = 1°C since it is the first layer below the ice
(ii) At Y = 4°C since it is the fourth layer below ice
APPEARS IN
संबंधित प्रश्न
1 kg of ice at 0°C is mixed with 1 kg of steam at 100°C. What will be the composition of the system when thermal equilibrium is reached? Latent heat of fusion of ice = 3.36 × 103 J kg−1 and latent heat of vaporization of water = 2.26 × 106 J kg−1.
On a winter day the temperature of the tap water is 20°C whereas the room temperature is 5°C. Water is stored in a tank of capacity 0.5 m3 for household use. If it were possible to use the heat liberated by the water to lift a 10 kg mass vertically, how high can it be lifted as the water comes to the room temperature? Take g = 10 m s−2.
A calorimeter contains 50 g of water at 50°C. The temperature falls to 45°C in 10 minutes. When the calorimeter contains 100 g of water at 50°C, it takes 18 minutes for the temperature to become 45°C. Find the water equivalent of the calorimeter.
Density of water is maximum at :
What do you understand by the anomalous expansion of water?
What is the criterion of choosing the two metals for a bimetal strip?
Write the name.
The instrument used to study anomalous behaviour of water.
Name the following diagram appropriately.

