Advertisements
Advertisements
प्रश्न
On a winter day the temperature of the tap water is 20°C whereas the room temperature is 5°C. Water is stored in a tank of capacity 0.5 m3 for household use. If it were possible to use the heat liberated by the water to lift a 10 kg mass vertically, how high can it be lifted as the water comes to the room temperature? Take g = 10 m s−2.
उत्तर
Given:-
Initial temperature of the water, Ti = 20°C
Final temperature of the water (room temperature), Tf = 5°C
Change in temeprature, ΔT = 20°C − 5°C = 15°C
Volume of water = 0.5 m3
Density of water, d = 1000 kg/m3
Mass of the water, M = (0.5 × 1000) kg = 500 kg
Heat liberated as the temperature of water changes from 20°C to 5°C is given by
Q = M×S×ΔT
Q = (500 × 4200 × 15) J
Q = (500 × 4200 × 15) J
Q = (75 × 420 × 1000) J
Q = 31500 × 1000 J = 315×105 J
Let the height to which the mass is lifted be h.
The energy required to lift the block = mgh = 10 × 10 × h = 100 h
Acording to the question,
Q = mgh
⇒ 100 h = 315×105 J
⇒ h = 315×103 m = 315 km
APPEARS IN
संबंधित प्रश्न
How does the anomalous expansion of water help aquatic organisms in cold climates?
Fill in the blank and rewrite the sentence.
The amount of water vapour in air is determined in terms of its ........... .
Observe the following graph. Considering the change in volume of water as its temperature is raised from 0°C, discuss the difference in the behaviour of water and other substances. What is this behaviour of water called?

Explain the following:
What is the role of the anomalous behaviour of water in preserving aquatic life in regions of cold climate?
Explain the following:
How can you relate the formation of water droplets on the outer surface of a bottle taken out of refrigerator with formation of dew?
A 50 kg man is running at a speed of 18 km h−1. If all the kinetic energy of the man can be used to increase the temperature of water from 20°C to 30°C, how much water can be heated with this energy?
Water is cooled from 4 °C to 0 °C. It will :
What do you understand by the anomalous expansion of water?
Explain why do vegetables and fruits get damaged during severe frost?
What are hot spots? How can you extract energy from a hot spot, if it does not come in contact with underground water?
What will be the approximate temperature of water in the lake shown in the following diagram?
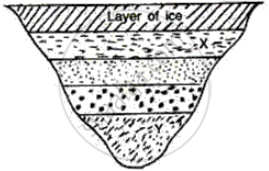
(i) At X, and
(ii) At Y?
Observe the given graph and answer the following questions:
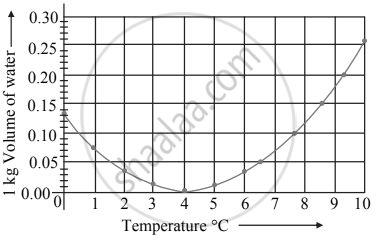
- Name the process represented in the figure.
- At what temperature does this process take place?
In cold regions, in winter the pipes for water supply break.
Name the following diagram appropriately.

A graph between the volume and temperature of water is shown. Explain the anomalous behaviour of water.

