Advertisements
Advertisements
प्रश्न
A dry pellet of a common base B, when kept in open absorbs moisture and turns sticky. The compound is also of chloralkali process. Identify B What type of reaction occurs when B is treated with an acidic oxide ? Write a balanced chemical equation for one such solution.
उत्तर
The available information suggests that the base B is sodium hydroxide (NaOH). It is a deliquescent substance and becomes sticky on absorbing moisture from atmosphere. It is commercially formed by the electrolysis of a strong solution of sodium chloride (brine).
It reacts with an acidic oxide such as CO2 or SO, gas to form corresponding salt and water. For example,
`2"NaOH" + "CO"_2 -> "Na"_2"CO"_3 + "H"_2"O"`
`2"NaOH" + "SO"_2 -> "Na"_2"SO"_3 + "H"_2"O"`
APPEARS IN
संबंधित प्रश्न
Sodium hydroxide is a ____________.
How will you protect yourself from the heat generated while diluting a concentrated acid?
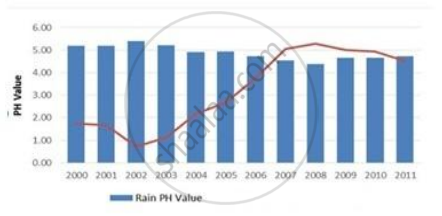
In which year is concentration of hydrogen ion the highest?
Which of the given options correctly represents the parent acid and base of Calcium Carbonate?
What will happen if we take dry HCl gas instead of the aqueous solution of HCl?
What will be the action of the following substances on blue litmus paper?
Dry HCl gas, Moistened NH3 gas, Lemon juice, Carbonated soft drink, Curd, Soap solution.
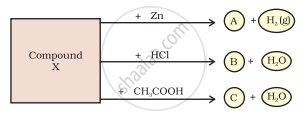
Identify the compound Y on the basis of the reactions given below. Also write the name and chemical formulae of A, B and C.
Assertion (A): It is advised that while diluting an acid one should add water to acid and not acid to water keeping the solution continuously stirred.
Reason (R): The process of dissolving an acid into water is highly exothermic.
Write the chemical equation for the following activity.
Dilute nitric acid was added to calcium oxide.
Four solutions, namely glucose, alcohol, hydrochloric acid and sulphuric acid filled in four separate beakers are connected one by one in an electric circuit with a bulb. The solutions in which the bulb will glow when current is passed are ______.
