Advertisements
Advertisements
प्रश्न
A dry pellet of a common base B, when kept in open absorbs moisture and turns sticky. The compound is also of chloralkali process. Identify B What type of reaction occurs when B is treated with an acidic oxide ? Write a balanced chemical equation for one such solution.
उत्तर
The available information suggests that the base B is sodium hydroxide (NaOH). It is a deliquescent substance and becomes sticky on absorbing moisture from atmosphere. It is commercially formed by the electrolysis of a strong solution of sodium chloride (brine).
It reacts with an acidic oxide such as CO2 or SO, gas to form corresponding salt and water. For example,
`2"NaOH" + "CO"_2 -> "Na"_2"CO"_3 + "H"_2"O"`
`2"NaOH" + "SO"_2 -> "Na"_2"SO"_3 + "H"_2"O"`
APPEARS IN
संबंधित प्रश्न
State the differences between acids and bases.
In terms of acidic strength, which one of the following is in the correct increasing order?
What is formed when zinc reacts with sodium hydroxide?
Sodium hydroxide is a ____________.
How would you distinguish between baking powder and washing soda by heating?
The change in colour of the moist litmus paper in the given set up is due to
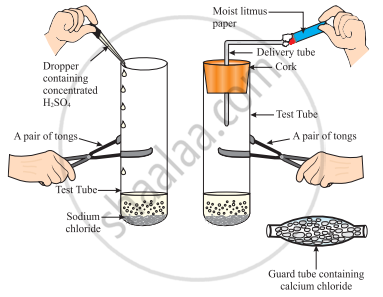
- presence of acid
- presence of base
- presence of H⁺(aq) in the solution
- presence of Litmus which acts as an indicator
Suggest a safe procedure of diluting a strong concentrated acid.
Write the chemical equation for the following activity.
Dilute nitric acid was added to calcium oxide.
Write the chemical equation for the following activity.
Dilute nitric acid was added to calcium oxide.
Four solutions, namely glucose, alcohol, hydrochloric acid and sulphuric acid filled in four separate beakers are connected one by one in an electric circuit with a bulb. The solutions in which the bulb will glow when current is passed are ______.
