Advertisements
Advertisements
प्रश्न
A heater supplies heat at the rate of 800 J/s. Find the time required to convert 50g of ice at -20°C into superheated steam at 140°C.
(Given: Specific heat of ice = 2.1 J/g, latent heat of ice = 340 J/gm, latent heat of steam = 2240 J/gm, specific heat of steam = 2.1 J/gm and specific heat of water = 4.2 J/gm.)
उत्तर
(i) Heat required to raise the temperature of ice from - 20°C to 0°C
= m × s × t = 50 × 2.1 × 20 = 2100 J
(ii) Heat required to melt ice at 0°C = mL = 50 × 340 = 1700 J
(iii) Heat required to heat water from °C to 100°C
= m × s × t = 50 × 4.2 × 100 = 21000 J
(iv) Heat required to convert water into steam at 100 C
= m × L = 50 × 2240 = 124000 J
(v) Heat required to heat steam from 100 C to 140 C
= m × s × t = 50 × 2.1 × 40 = 4200 J
Total quantity of heat = (2100 + 17000 + 21000 + 112000 + 4200) J
= 156300 J
If t is the time taken by heater, in second.
Thewn 800 × t = 156300 J
∴ t = `156300/800`s = 195.375 s = 195.4s
APPEARS IN
संबंधित प्रश्न
What is the other name given to the principle of the mixtures?
Define calorimetry ?
Explain the melting point ?
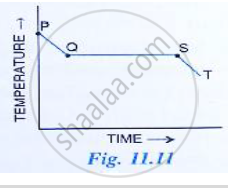
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
The amount of heat energy required to convert 1 kg of ice at – 10℃ to water at 100℃ is 7,77,000 J. Calculate the specific latent heat of ice. Specific heat capacity of ice = 2100 J kg-1K-1, Specific heat capacity of water = 4200 J kg-1 K-1.
A block of lead of mass 250 g, at 27°C was heated in a furnace till it completely melted. Find the quantity of heat required
(a) To bring the lead block upto its melting point,
(b) To completely melt the block at its melting point (Melting point of lead is 327°C, Sp. heat cap. of lead is 0.13 J/gk and latent heat of fusion of lead is 26 J/g)
Fill in the following blank using suitable word:
Whenever mechanical work is done, .......... Is produced.
Why does the heat supplied to a substance during its change of state not cause any rise in its temperature?
Explain briefly, how you would determine the specific heat capacity of a liquid?
A mass of 40g of brass of specific heat capacity 0.85 Jg-1 K-1 is heated in an oven and then quickly transferred into 240g of water at 30°C in a calorimeter of mass 60g and specific heat capacity 0.4 Jg-1 K-1. If the final temperature is 50°C. What was the temperature of the oven?
