Advertisements
Advertisements
प्रश्न
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
उत्तर
Incase of o-nitrophenol Intramolecular hydrogen bonding is there which is weaker than intermolecular hydrogen bonding in p-nitrophenol. Hence o-nitrophenol is steam volatile.
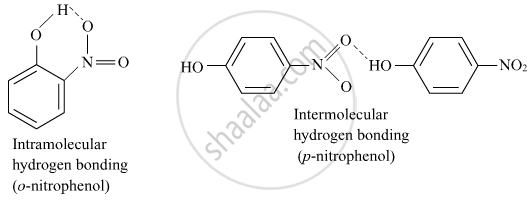
APPEARS IN
संबंधित प्रश्न
Give two reactions that show the acidic nature of phenol.
Intermolecular hydrogen bonding is strongest in ______.
Phenols do not react with one of the following:
The ionization constant of phenol is higher than that of ethanol because ____________.
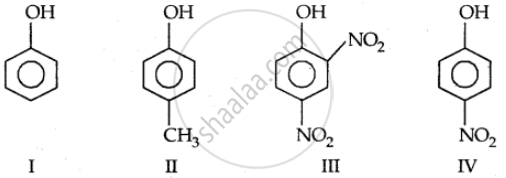
Strength of acidity is in order:
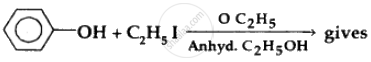
What is the correct order of reactivity of alcohols in the following reaction?
\[\ce{R-OH + HCl ->[ZnCl2] R-Cl + H2O}\]
Phenol is used in the manufacture of
Arrange the following in the increasing order of their property indicated:
4-Nitrobenzoic acid, benzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxy benzoic acid (Acid strength)
Which one of the following has the lowest pKa value?
