Advertisements
Advertisements
Question
Account for the following:
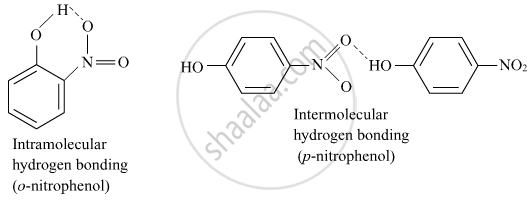
o-nitrophenol is more steam volatile than p-nitrophenol.
Solution
Incase of o-nitrophenol Intramolecular hydrogen bonding is there which is weaker than intermolecular hydrogen bonding in p-nitrophenol. Hence o-nitrophenol is steam volatile.
APPEARS IN
RELATED QUESTIONS
The ionization constant of phenol is higher than that of ethanol because ____________.
In the following compounds:
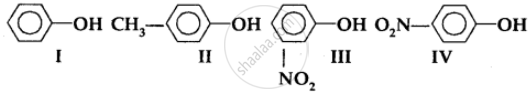
The order of acidity is
In CH3CH2OH, the bond that undergoes heterolytical change most readily is ____________.
Strength of acidity is in order:
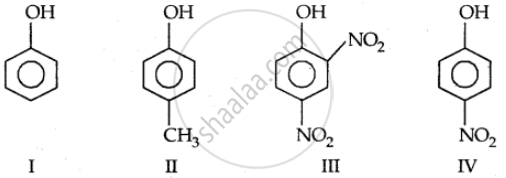
Which of the following statements is true:
Assertion: o-Nitrophenol is less soluble in water than the m- and p-isomers.
Reason: m- and p- Nitrophenols exist as associated molecules.
Which one of the following has the lowest pKa value?
Give the structure of the product you would expect when the following alcohol reacts with HCl–ZnCl2.
2-Methylbutan-2-ol
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
Butan-1-ol
Compare acidity of phenol with that of ethanol.
