Advertisements
Advertisements
प्रश्न
Although geometries of NH3 and H2O molecules are distorted tetrahedral, bond angle in water is less than that of ammonia. Discuss.
उत्तर १
The molecular geometry of NH3 and H2O can be shown as:

The central atom (N) in NH3 has one lone pair and there are three bond pairs. In H2O, there are two lone pairs and two bond pairs.
The two lone pairs present in the oxygen atom of H2O molecule repels the two bond pairs. This repulsion is stronger than the repulsion between the lone pair and the three bond pairs on the nitrogen atom.
Since the repulsions on the bond pairs in H2O molecule are greater than that in NH3, the bond angle in water is less than that of ammonia.
उत्तर २
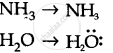
Because of two lone pairs of electrons on O-atom, repulsion on bond pairs is greater in H20 in comparison to NH3 . Thus, the bond angle is less in H20 molecules
APPEARS IN
संबंधित प्रश्न
Discuss the shape of the following molecules using the VSEPR model:
BeCl2, BCl3, SiCl4, AsF5, H2S, PH3
Select and write the most appropriate alternatives from the given choices.
Valence Shell Electron Pair repulsion (VSEPR) theory is used to predict which of the following:
Shape of ClF3 is ______.
Explain VSEPR theory. Applying this theory to predict the shapes of IF7 and SF6.
The H-N-H bond angle in NH3 molecule is ____________.
Select the INCORRECT match.
Stable form of A may be represented by the formula:
Elements \[\ce{X, Y}\] and \[\ce{Z}\] have 4, 5 and 7 valence electrons respectively. Write the molecular formula of the compounds formed by these elements individually with hydrogen.
Elements \[\ce{X, Y}\] and \[\ce{Z}\] have 4, 5 and 7 valence electrons respectively. Which of these compounds will have the highest dipole moment?
Which of the possible molecule/species is having maximum values for dipole moment. (where "A" is the central atom)?
Consider the species CH4, `"NH"_4^+` and `"BH"_4^-`. Choose the correct option with respect to these species.
Number of lone pair(s) of electrons on central atom and the shape of BrF3 molecule respectively are ______.
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: The H-O-H bond angle in water molecule is 104.5°.
Reason R: The lone pair-lone pair repulsion of electrons is higher than the bond pair-bond pair repulsion.
In the light of the above statements, choose the correct answer from the options given below:
What is the geometry of a water molecule?
What is the number of lone pair of electrons in IF7?
Identify compound having square pyramidal-shape:from following.
