Advertisements
Advertisements
प्रश्न
Aluminium is extracted from its chief ore bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Mention one reason for the use of aluminium in thermite welding. Which particular property of cast iron makes it unsuitable for the construction of bridges?
उत्तर
As a reducing agent: Aluminium has a high affinity for oxygen. It readily removes oxygen from oxides of less reactive metals.
APPEARS IN
संबंधित प्रश्न
For cryolite, explain its significance in the extraction of aluminium.
In order to obtain 1 tonne of aluminium, the following inputs are required: 4 tonnes of bauxite, 150 kg of sodium hydroxide and 600 kg of graphite. The aluminium compound in bauxite is aluminium oxide and the main impurity is iron (III) oxide. Aluminium is obtained by the electrolysis of aluminium oxide dissolved in cryolite.
- Write the formula of cryolite.
- Write down the word which correctly completes the following sentence.
By dissolving aluminium oxide in cryolite, a ______ (conducting/non-conducting) solution is produced. - Why is so much graphite required for the electrolytic process?
- Write the equation for the reaction which takes place at the cathode.
- What is cathode made up of?
| Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction. |
Name a chemical used for dissolving aluminium oxide. In which state is the chemical used?
Why is food containing iron salts should not be cooked in aluminium utensils?
Describe the role played in the extraction of aluminum : Graphite
Explain why :
Iron sheets are coated with zinc during galvanization.
Name the alloy used for the following purpose.
Surgical instruments
Answer the following question:
Write the chemical formula of one main ore of iron and aluminium.
State the relevant reason for the following:
A layer of powered coke is used over the electrolytic mixture in Hall Heroult’s process.
The following sketch illustrates the process of conversion of Alumina to Aluminium:
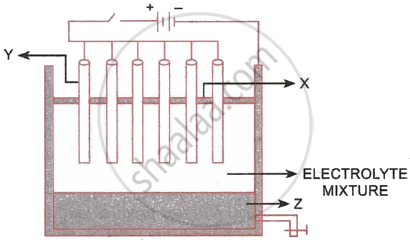
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
