Advertisements
Advertisements
प्रश्न
An element A which is a part of common salt and kept under kerosene reacts with another element B of atomic number 17 to give a product C. When an aqueous solution of product C is electrolysed then a compound D is formed and two gases are liberated.
(a) What are A and B?
(b) Identify C and D.
(c) What will be the action of C on litmus solution? Why?
(d) Write formula of the compound formed when element B reacts with an element E having atomic number 5.
उत्तर
(a) Element A is sodium and element B is chlorine.
Sodium is kept under kerosene because it is highly reactive. Element with atomic number 17 is chlorine. Thus, element A is sodium and element B is chlorine.
(b) When element A, sodium, reacts with element B, chlorine, it forms a product C, sodium chloride, NaCl.
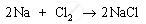
When aqueous solution of sodium chloride is electrolysed, compound D, sodium hydroxide (NaOH), is formed along with the evolution of hydrogen and chlorine gases.
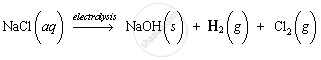
(c) Compound C is sodium chloride. It has no action on litmus solution. Because sodium chloride is a salt, it is neutral in nature and does not show any action with litmus solution.
(d) Element B is chlorine. It is in gaseous state at room temperature.
(e) The element E with atomic number 5 is boron. When element B, chlorine, reacts with element E, boron, it forms a compound boron trichloride. The formula of the compound formed is BCl3.
2B + 3Cl2 → 2BCl3
APPEARS IN
संबंधित प्रश्न
The process by which sulphide ore is concentrated.
Name the gas evolved when cinnabar ore is roasted.
Describe the extraction of zinc metal from its sulphide ore (zinc blende). Write equations of the reactions involved.
Which of the following is an ore of mercury metal?
(a) rock salt
(b) cinnabar
(c) calamine
(d) haematite
During galvanisation, iron metal is given a thin coating of one of the following metals. This metal is:
(a) chromium
(b) tin
(c) zinc
(d) copper
The following questions relate to the extraction of aluminium by electrolysis.
Explain why it is necessary to renew the anode periodically.
Choose the correct answer from the options given below:
The metal is a liquid at room temperature.
Write the name.
The process of strong heating of carbonate ores in insufficient air–
Explain the reactions of nonmetals with water with the help of examples.
Explain the following reaction with the balanced equation.
Zinc oxide is treated with carbon
