Advertisements
Advertisements
प्रश्न
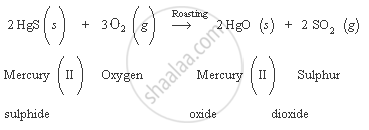
Name the gas evolved when cinnabar ore is roasted.
उत्तर
Sulphur dioxide gas is evolved when cinnabar ore is roasted.
APPEARS IN
संबंधित प्रश्न
M is a metal above hydrogen in the activity series and its oxide has the formula M2O. This oxide when dissolved in water forms the corresponding hydroxide which is a good conductor of electricity. In the above context, answer the following:
1) What kind of combination exists between M and O?
2)How many electrons are there in the outermost shell of M?
3) Name the group to which M belongs.
4) State the reaction taking place at the cathode.
5) Name the product at the anode.
A zinc ore gave CO2 on treatment with a dilute acid. Identify the ore and write its chemical formula.
Why are the metals like Na, K, Ca and Mg never found in their free state in nature?
How is aluminium metal extracted? Explain with the help of an equation.
Define the following term.
Gangue
Give the chemical formula of :
Sodium aluminate
Find the odd one out and give its explanation.
Write the name.
Metals which are amphoteric in nature.
Explain the following reaction with the balanced equation.
Sodium aluminate reacts with water
Two ores A and B were taken. On heating ore A gives CO2 whereas, ore B gives SO2. What steps will you take to convert them into metals?
