Advertisements
Advertisements
प्रश्न
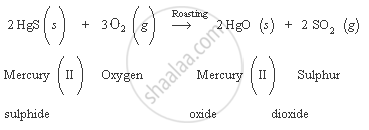
Name the gas evolved when cinnabar ore is roasted.
उत्तर
Sulphur dioxide gas is evolved when cinnabar ore is roasted.
APPEARS IN
संबंधित प्रश्न
Which metal becomes black in the presence of hydrogen sulphide gas in air?
Explain why, carbon cannot reduce oxides of sodium or magnesium.
Why are the metals like Na, K, Ca and Mg never found in their free state in nature?
Name two metals which occur in nature in free state as well as in combined state.
Calamine ore can be used to extract one of the following metals. This metal is:
(a) copper
(b) mercury
(c) aluminium
(d) zinc
Distinguish between ‘roasting’ and ‘calcination’. Which of these two is used for sulphide ores and why?
Name the following:
Two metals which are found in the free or native state.
Extraction of moderately reactive elements is done by _______ and _______ method.
Electrical wires have a coating of an insulting material. The material, generally used is
A mineral from which the metal can be extracted economically and conveniently is known as ______.
