Advertisements
Advertisements
Question
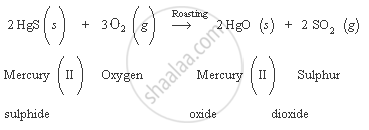
Name the gas evolved when cinnabar ore is roasted.
Solution
Sulphur dioxide gas is evolved when cinnabar ore is roasted.
APPEARS IN
RELATED QUESTIONS
What chemical process is used for obtaining a metal from its oxide?
Fill in the following blank with suitable word:
Bronze is an alloy of copper and .........
Define the term ore.
Name one ore of sodium. Name the sodium compound present in this ore and write its chemical formula.
Name the metal which is extracted from the ore called 'rock salt'.
A sulphide ore is converted into metal oxide by the process of:
(a) carbonation
(b) roasting
(c) calcination
(d) anodising
Give the principles of the froth floatation process.
Give reasons, why aluminum is used in:
painting electric and telegraphic poles
Write the name.
Metals which are amphoteric in nature.
2 mL each of concentrated HCl, HNO3 and a mixture of concentrated HCl and concentrated HNO3 in the ratio of 3 : 1 were taken in test tubes labelled as A, B and C. A small piece of metal was put in each test tube. No change occurred in test tubes A and B but the metal got dissolved in test tube C respectively. The metal could be
