Advertisements
Advertisements
प्रश्न
An empty glass bottle is fitted with a narrow tube at its mouth. The open end of the tube is kept in a beaker containing water. When the bottle is heated, bubbles of air are seen escaping into water. Explain the reason.
उत्तर
When the bottle is heated, the air in it expands and the solubility of gases in it decreases which escapes from water in the form of bubbles.
APPEARS IN
संबंधित प्रश्न
What do you understand by thermal expansion of a substance?
Two iron rods – one 10 m long and the other 5 m long, are heated to the same rise in temperature. Which will expand more?
Why is one end of a steel girder in a bridge kept on rollers instead of fixing it in pillar?
A metal plate is heated. State three factors on which the increase in its area will depend.
A cubical metal solid block is heated. How will its volume change?
State one application of thermal expansion of liquids.
An iron washer is heated. State the effect on its internal diameter.
At what temperature the pure water has (a) maximum density (b) minimum volume?
The diagram shows a sketch graph showing a change in volume of a given mass of ice at -5°C to water at 15°C.
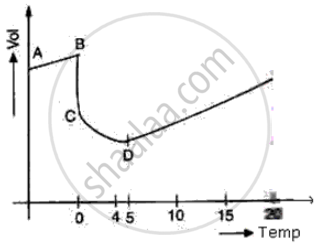
(i) Why does the temperature along BC remain constant?
(ii) When is the volume of water minimum?
(iii) At what temperature does water have maximum density? Explain.
- Assertion: Gas is highly compressible than solid and liquid
- Reason: Interatomic or intermolecular distance in the gas is comparably high.
