Advertisements
Advertisements
प्रश्न
Answer the following.
Why the elements belonging to the same group do have similar chemical properties?
उत्तर
- Chemical properties of elements depend upon their valency.
- Elements belonging to the same group have the same valency.
Hence, the elements belonging to the same group show similar chemical properties.
APPEARS IN
संबंधित प्रश्न
Explain the following.
The ionic radii of FΘ and Na⊕ are 133 and 98 pm, respectively.
Explain the following.
13Al is a metal, 14Si is a metalloid and 15P is a nonmetal.
Explain the following.
Cu forms coloured salts while Zn forms colourless salts.
Answer the following.
Explain the screening effect with a suitable example.
Choose the correct option.
Consider the elements B, Al, Mg, and K predict the correct order of metallic character:
Choose the correct option.
The lanthanides are placed in the periodic table at
Choose the correct option.
Which of the following pairs is isoelectronic?
Answer the following question.
For the following pair, indicate which of the two species is of large size:
Mg2+ or Ca2+
Answer the following question.
Select the smaller ion form the following pair:
K+, Li+
Answer the following question.
Select the smaller ion form the following pair:
N3–, F–
With the help of a diagram answer the questions are given below:
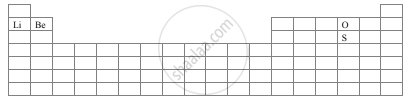
- Which atom should have smaller ionization energy, oxygen, or sulphur?
- The lithium forms +1 ions while beryllium forms +2 ions?
Define ionic radius
Define electronegativity
What are the valence electrons?
Define ionization enthalpy.
Name the factors on which ionization enthalpy depends?
How does ionization enthalpy vary down the group and across a period?
How the atomic size vary in a group and across a period? Explain with suitable example.
Give reason.
Inert gases have exceptionally high ionization enthalpies.
Give reason.
Fluorine has less electron affinity than chlorine.
Give reason.
Noble gases possess relatively large atomic size.
